
Mind & Matter
259 episodes — Page 1 of 6

Opioid Addiction: RNA Biology, Brain Inflammation & Psychedelic Therapy
This is a free preview of a paid episode. To hear more, visit mindandmatter.substack.comWide release: February 19, 2026. Not medical advice.TOPICS DISCUSSED:* Gene regulation basics: DNA transcribes to RNAs, including non-coding types like microRNAs that inhibit mRNA translation into proteins, influencing up to 60% of the proteome.* Non-coding RNAs in neuroplasticity: MicroRNAs and circular RNAs regulate synaptic changes, with activity-induced ones like miR-485-5p linked to rapid responses in drug cue memory and addiction reinforcement.* Opioid addiction models: Rats self-administer heroin or fentanyl via levers, showing compulsive seeking; fentanyl’s higher potency drives faster learning but similar long-term effects to heroin when doses are equated.* Differences between opioids: Heroin and fentanyl both activate mu-opioid receptors for euphoria and dopamine release, but fentanyl lingers longer; no major behavioral differences in seeking once potency is matched.* Psilocybin’s effects on addiction: A single psilocybin dose post-abstinence reduces heroin-seeking in rats by dampening neuroinflammation in brain regions like the nucleus accumbens and prefrontal cortex.* Brain Inflammation: Opioids induce pro-inflammatory changes via cytokines like IL-17A and pathways like TNF-alpha, leading to glial activation and blood-brain barrier leaks; psilocybin counters this.* MicroRNA biomarkers: Blood microRNAs reflect gene expression patterns tied to disease states, with potential to predict opioid relapse risk, treatment response, or neonatal withdrawal severity non-invasively.* Future research: Ongoing work links psilocybin’s serotonin 2A activation to anti-inflammatory gene changes, plus human studies on microRNAs for personalized addiction treatments.ABOUT THE GUEST: Stephanie Daws, PhD is an associate professor at Temple University in the Center for Substance Abuse Research and Department of Neurosciences, where she researches mechanisms of drug-seeking behavior with a focus on opioids and psychedelics.RELATED EPISODE:* M&M 2 | Psilocybin, LSD, Ketamine, Inflammation & Novel Psychedelic Medicines | Charles NicholsListen or watch on your favorite platform:* Audio version: [Apple] [Spotify] [Elsewhere]* Video version: [YouTube]* Support M&M if you find value in this content.PRACTICAL TAKEAWAYS:* For those in recovery, discuss medication options like buprenorphine or methadone with providers, as individual responses vary and biomarkers may one day guide choices.* Be aware of fentanyl’s extreme potency in street drugs, which heightens overdose risk.* Explore emerging psychedelic therapies under medical supervision, as psilocybin shows promise in reducing drug cravings via plasticity and inflammation modulation.SUBSCRIBER CONTENT BELOW: Reference paper + episode transcript.

Estrogen & Body Fat: Menopause, Puberty, Females vs. Males
This is a free preview of a paid episode. To hear more, visit mindandmatter.substack.comWide release: February 14, 2026. Not medical advice.TOPICS DISCUSSED:* Adipose tissue basics: White fat primarily stores energy in large lipid droplets, while brown fat burns fatty acids for heat via high mitochondrial density; white fat can “brown” with exercise or certain foods like capsaicin.* Fat distribution & health: Subcutaneous fat (under skin) is more insulin-sensitive and less problematic than visceral fat (around organs), which links to metabolic issues; females store more subcutaneously pre-menopause, shifting to visceral post-menopause.* Estrogen signaling: Estradiol binds nuclear and membrane receptors to regulate gene expression and mitochondrial function; it enhances insulin sensitivity and browning in fat cells, with receptors like ER-alpha feminizing fat distribution.* Fat storage: Fat cells enlarge (hypertrophy) more than multiply in obesity, leading to hypoxia, inflammation, and insulin resistance; excess fatty acids spill to liver and muscle, worsening metabolic dysfunction.* Menopause effects: Estrogen drop causes visceral fat gain, reduced energy expenditure, insulin resistance, and higher metabolic disease risk; symptoms include hot flashes and reduced exercise motivation, modeled in rodents via ovary removal.* Exercise & estrogen links: Exercise boosts estrogen receptor expression and mitochondrial density in fat, mimicking estrogen’s browning effects; synergism may explain reduced exercise responsiveness post-menopause.* Brain-fat connections: Estrogen in the nucleus accumbens influences exercise motivation and fat browning; manipulations there alter running behavior and adipose metabolism in rodents.ABOUT THE GUEST: Victoria Vieira-Potter, PhD leads a lab at the University of Missouri, studying how estrogen and exercise influence adipose cells.RELATED EPISODE:* M&M 174 | Adipose Tissue & Body Fat: Obesity, Insulin, Leptin, Fertility, Weight Loss & GLP-1 Drugs | Sean HartigListen or watch on your favorite platform:* Audio version: [Apple] [Spotify] [Elsewhere]* Video version: [YouTube]* Support M&M if you find value in this content.PRACTICAL TAKEAWAYS:* Regular exercise increases mitochondrial function and insulin sensitivity in fat cells, potentially aiding weight management and metabolic health for both sexes.* Post-menopausal women may benefit from maintaining physical activity to counter reduced energy expenditure and visceral fat gain, possibly buffering menopause symptoms.* Hormone therapy could improve metabolic outcomes like insulin sensitivity but requires physician consultation to weigh potential risks such as cancer.* Building lean muscle mass through resistance training supports basal metabolic rate as estrogen declines with age.SUBSCRIBER CONTENT BELOW: Reference paper + episode transcript.

The Claustrum: Cognition, Consciousness, Alcohol & Psychedelics | 278
This is a free preview of a paid episode. To hear more, visit mindandmatter.substack.comWide release: February 9, 2026. Not medical advice.TOPICS DISCUSSED:* Cerebral cortex structure: Described as a six-layered structure with pyramidal neurons and inhibitory interneurons; information flows between layers and regions to process sensory input and enable complex behaviors.* Claustrum anatomy & connectivity: A sheet-like subcortical structure embedded in white matter, bidirectionally connected to cortical areas, especially prefrontal regions in rodents, with broader connections in primates and humans suggesting an integrative role.* Claustrum function in cognition: Experiments show claustrum activation during task switches from easy to demanding modes, synchronizing cortical networks via inhibition and rebound excitation, potentially enabling flexible behavior.* Mouse models in neuroscience: Mice are used for genetic tractability to manipulate and monitor specific circuits, revealing claustrum’s role in vigilance tasks but not simple ones.* Alcohol’s effects on brain circuits: Chronic alcohol promotes inflexible behaviors by altering striatal interneurons and inhibitory inputs, leading to compulsive drinking despite aversive consequences.* Psychedelics & brain networks: Psilocybin disrupts default mode and other networks, inhibits claustrum via serotonin 1B receptors, with effects persisting 24 hours, possibly contributing to therapeutic benefits.* Evolution of claustrum: Connectivity expands from rodents to humans, shifting from cognitive-specific to broader network control, including anti-correlated states like default mode versus task-engaged.* Integration of claustrum & basal ganglia: Claustrum funnels prefrontal signals to basal ganglia for action selection; alcohol may impair this, exacerbating inflexibility in addiction.ABOUT THE GUEST: Brian Mathur, PhD is a professor in the Department of Pharmacology and Physiology at the University of Maryland School of Medicine, where he leads a neuroscience lab studying brain circuits underlying flexible and inflexible behaviors using mouse models, with a focus on alcohol use disorder.RELATED EPISODE:* M&M 226 | Psychedelics & Cerebral Cortex: Neuroplasticity, Psilocybin, Ketamine | Alex Kwan* Full audio version: [Apple] [Spotify] [Elsewhere]* Full video version: [YouTube]* Support M&M if you find value in this content.PRACTICAL TAKEAWAYS:* Moderate alcohol consumption can impair decision-making and coordination by affecting prefrontal cortex and cerebellum, so limit intake during tasks requiring focus.* Chronic heavy drinking may lock in maladaptive habits; seeking therapy or support can help regain behavioral flexibility.* Psychedelics like psilocybin may reset rigid thought patterns in conditions like depression, but use under medical supervision for potential long-term benefits.SUBSCRIBER CONTENT BELOW: Reference paper + episode transcript.

Psychedelic Neurobiology: Sex-Specific Effects of MDMA & Psilocybin in Addiction & Reward Behavior
This is a free preview of a paid episode. To hear more, visit mindandmatter.substack.comWide release: February 4, 2026. Not medical advice.TOPICS DISCUSSED:* Biased agonism: Different drugs activate the same receptor (e.g., 5-HT2A) but trigger varied intracellular pathways, explaining why LSD is psychedelic while similar lisuride is not.* Enantiomers & isomers: Mirror-image versions of drugs like MDMA (S and R forms) and LSD (four isomers) often produce distinct effects; only one LSD isomer is psychedelic, for example.* MDMA isomer effects: S-MDMA induces stronger head twitches (psychedelic proxy) via serotonin release, and increases dendritic spines in male mice but not females; R-MDMA has somewhat different effects.* Sex-specific drug responses: In mice, females show stronger psychedelic effects (head twitches) from psilocybin and DOI at the same dose, but males exhibit greater post-acute benefits like reduced opioid withdrawal.* Psilocybin in opioid addiction models: A single dose reduces place preference for oxycodone and withdrawal symptoms in male mice more than females, with opposite spine density effects in reward-related brain areas.* Mechanisms beyond 5-HT2A: Psychedelics involve other receptors like metabotropic glutamate receptors, forming dimers with 5-HT2A to enable dual signaling pathways; effects in subcortical regions like nucleus accumbens are 5-HT2A-independent.* Clinical implications: Street MDMA may vary in S/R ratios, affecting experiences; clinical trials often use racemic mixtures without weight-adjusted dosing, potentially missing sex differences.ABOUT THE GUEST: Javier Gonzalez-Maeso, PhD is a professor of pharmacology and toxicology at Virginia Commonwealth University, with a PhD in medicine from Spain focused on G-protein coupled receptors and human brain studies in depression and addiction.RELATED EPISODE:* M&M 230 | Psilocybin & MDMA: Inflammation, Stress & Brain-Body Communication | Michael Wheeler* Full audio version: [Apple] [Spotify] [Elsewhere]* Full video version: [YouTube]* Support M&M if you find value in this content.SUBSCRIBER CONTENT BELOW: Practical takeaways, reference paper, episode transcript.

AMPK: Biochemistry of Nutrient Sensing, Fasting, Cell Repair & Growth | Greg Steinberg
This is a free preview of a paid episode. To hear more, visit mindandmatter.substack.comWide release: January 25, 2026. Not medical advice.TOPICS DISCUSSED:* Cellular energy basics: ATP/ADP/AMP as energy currencies; AMP kinase activates on low ATP to conserve and redirect resources, like a budget manager.* AMP kinase mechanics: Heterotrimeric enzyme phosphorylating 100+ substrates; localizes dynamically in cytosol, mitochondria, nucleus to integrate signals.* Nutrient detection: Senses fatty acids in fasting/ketogenic states, boosting fat oxidation and mitochondria independent of energy drops.* Fasting/exercise impacts: Elevate AMP kinase for mitophagy, better fuel switching; mimic historical scarcity absent under modern constant feeding.* mTOR relationship: AMP kinase inhibits mTOR to stop growth in low energy states; feedback loop disrupted by abundance, promoting tissue buildup.* Disease links: Low AMP kinase in obesity/diabetes reduces flexibility; activation prevents cancer but may aid tumor survival in therapy.* Drugs/diets: Metformin and GLP-1s like Ozempic activate AMP kinase for glucose control/weight loss; ketogenic diets activate at intermediate levels for fat efficiency.ABOUT THE GUEST: Gregory Steinberg, PhD is a Professor of Medicine at McMaster University and co-directs the Centre for Metabolism, Obesity, and Diabetes Research, focusing on cellular energy sensors like AMP kinase.RELATED EPISODE:* M&M 260 | Energy Resistance Principle in Life, Healing & Disease | Martin Picard & Nirosha Murugan* Full audio version: [Apple] [Spotify] [Elsewhere]* Full video version: [YouTube]* Support M&M if you find value in this content.SUBSCRIBER CONTENT BELOW: Practical takeaways, reference paper, episode transcript.

Farm Food: Soy in Animal Feed, Pesticides, Phytoestrogens, Seed Oils & Regenerative Farming
This is a free preview of a paid episode. To hear more, visit mindandmatter.substack.comWide release: January 19, 2026. Not medical advice.TOPICS DISCUSSED:* Soy’s rise in farming: Originating from paint uses and wartime chemicals, soy became dominant due to subsidies and large-scale farming practices, but introduces high PUFAs, phytoestrogens, and pesticides into animal feeds.* Feed’s impact on eggs: Chicken feeds high in soy and corn raise linoleic acid levels in eggs compared to low-soy alternatives; low-PUFA eggs improve digestibility for many.* PUFAs & health effects: Feeding PUFAs fattens animals faster and disrupts human gut lining and metabolism; body fat composition changes take time, linking to chronic issues like low energy.* Phytoestrogens in soy: These compounds pass into animal products, affecting estrogen signaling and gut health; mainstream nutrition often overlooks their risks despite historical low exposure.* Dairy variations: Raw milk retains lactoferrin for iron regulation and gut support, plus enzymes and probiotics destroyed in pasteurization; it may resolve dairy intolerances for some.* Regenerative farming model: Armstrong’s cooperative emphasizes traditional feeds to “resaturate” foods, reducing PUFAs; supports small farms amid declining farm numbers.* Ancestral diets insight: Traditional diets vary but share low PUFA levels, avoiding modern chronic illnesses tied to industrial fats.ABOUT THE GUEST: Ashley Armstrong, PhD co-founded Angel Acres Farm in Michigan and now runs Nourish Food Club, a cooperative of small farms producing low-PUFA, soy-free animal foods.RELATED EPISODE:* M&M 273: Nutrition Content of Animal & Plant Foods: Beef, Plant-Based Meat, Raw vs. Processed Milk* Full audio version: [Apple] [Spotify] [Elsewhere]* Full video version: [YouTube]* Support M&M if you find value in this content.SUBSCRIBER CONTENT BELOW: Practical takeaways, reference paper, episode transcript.
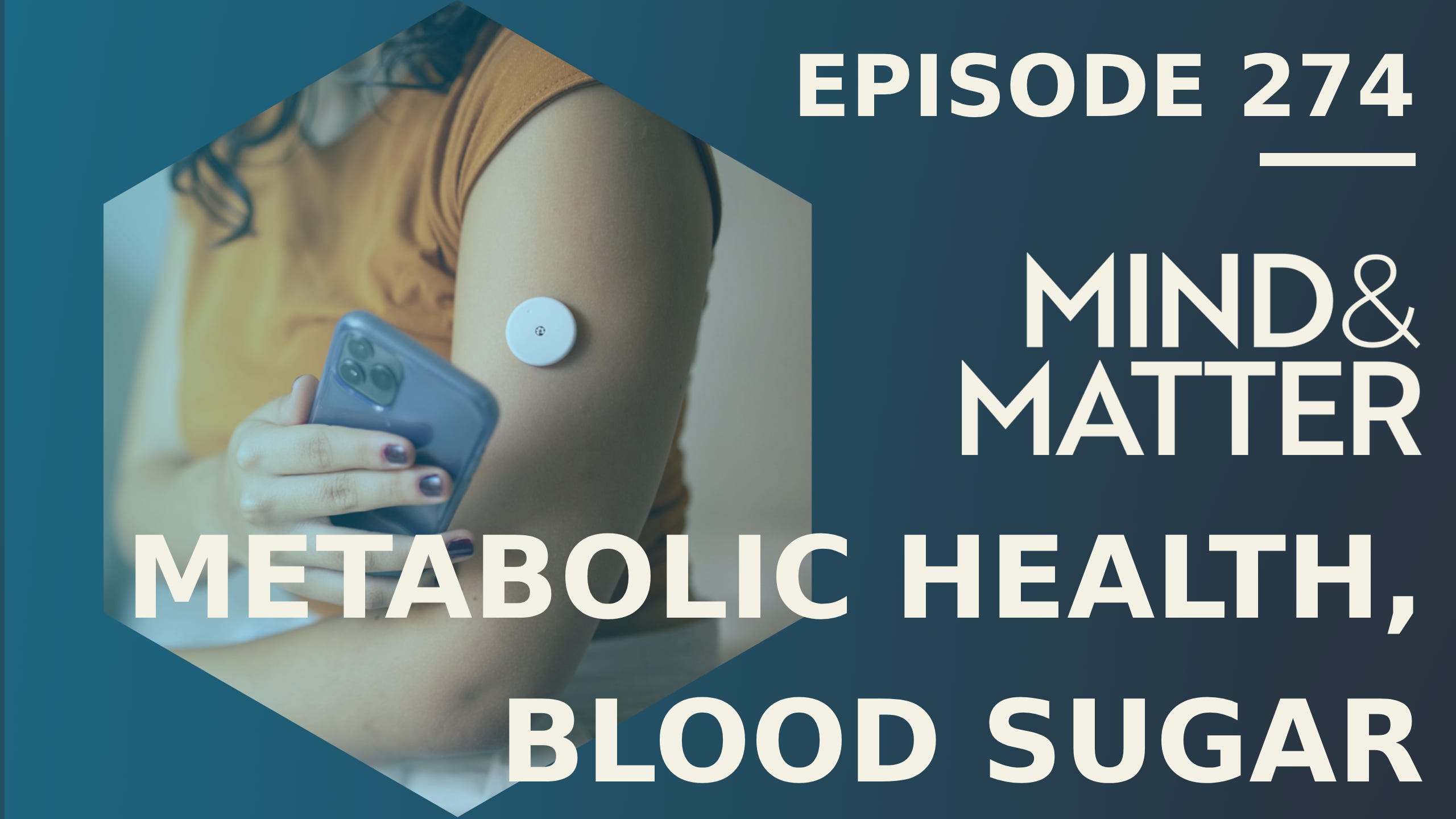
Personalized Nutrition: Individual Responses to Foods, Fiber, Fats & Maternal Health
This is a free preview of a paid episode. To hear more, visit mindandmatter.substack.comWide release: January 14, 2026. Not medical advice.TOPICS DISCUSSED:* Personalized health profiling: Snyder’s lab sequences genomes and measures molecules from blood, urine, and microbiomes to detect diseases early, emphasizing data-driven discovery over assumptions.* Fiber variations and effects: Responses to dietary fibers vary individually, with microbiome playing a key role.* Glycemic responses to carbs: People spike differently to foods like rice, potatoes, or bread; tied to insulin resistance subtypes in muscles, liver, or pancreas.* Type 2 diabetes heterogeneity: 90% of diabetes cases are type 2 with subtypes like muscle or hepatic resistance; triggers include genetics and infections, with prediabetes often undetected.* Continuous glucose monitors (CGMs): Affordable tools measure glucose every five minutes, revealing personal food triggers; spikes link to risks like cardiovascular disease and cancer.* Lipidomics: Lipids are diverse, serving as energy sources, structural components, and signaling molecules; understudied but key in metabolism and gene expression.* Maternal health & pregnancy: Metabolomics shows thousands of changes, including hormone shifts; cord blood analysis reveals fetal nutrient use and drug transfers, with markers for preterm birth or healthy outcomes.* Future research: Exploring epigenetic changes in diabetes, personalized nutrition mitigation, and digital markers from wearables for mental health tied to diet and gut-brain axis.ABOUT THE GUEST: Michael Snyder, PhD is the Stanford W. Ascherman Professor of Genetics and former chair of the Department of Genetics at Stanford University School of Medicine, where he directs the Center for Genomics and Personalized Medicine.RELATED EPISODE:* M&M 205: Systems Biology, Personalized Medicine, AI & the Future of Health | Lee Hood* Full audio version: [Apple] [Spotify] [Elsewhere]* Full video version: [YouTube]* Support M&M if you find value in this content.SUBSCRIBER CONTENT BELOW: Practical takeaways, reference paper, episode transcript.

Nutrition Content of Animal & Plant Foods: Beef, Plant-Based Meat, Raw vs. Processed Milk
This is a free preview of a paid episode. To hear more, visit mindandmatter.substack.comWide release: January 9, 2026. Not medical advice.TOPICS DISCUSSED:* Nutrient density profiling: Labs analyze thousands of compounds beyond macros and vitamins; this “dark matter” includes phytonutrients that may support health despite not being essential.* Phytonutrients in foods: Plant secondary metabolites like polyphenols act as antioxidants and influence pathways like mTOR; animals convert plant compounds into bioactives humans access via meat.* Red meat definition: Refers to meats high in myoglobin, including beef and lamb; most meats are red in wild forms, but human intervention affects color and classification.* Ruminant animals: Animals like cows that have multi-chambered stomachs to digest plants; this metabolism differs from non-ruminants (e.g. chickens), affecting nutrient profiles in their meat.* Grass-fed vs. grain-fed beef: Grass-fed has higher omega-3 fats and phytonutrients from diverse plants; studies show it improves human omega-6:3 ratios and biomarkers from grass-fed, pastured-raised animals.* Farming practices & variations: Plant diversity boosts beef nutrients.* Upcoming research: Long-term trials on effects of pasture-raised foods on human health; interactive dashboards for farmers to profile nutrients and inform policy.ABOUT THE GUEST: Stephan Van Vliet, PhD is an Assistant Professor in the Department of Nutrition, Dietetics, and Food Sciences at Utah State University, where he directs the Center for Human Nutrition Studies, focusing on linking food production systems to nutrient profiles and conducting clinical trials on health impacts.RELATED EPISODE:* M&M 106: Diet, Macronutrients, Micronutrients, Taste, Whole vs. Processed Food, Obesity & Weight Loss, Comparative Biology of Feeding Behavior | Stephen Simpson & David Raubenheimer * Full audio version: [Apple] [Spotify] [Elsewhere]* Full video version: [YouTube]* Support M&M if you find value in this content.SUBSCRIBER CONTENT BELOW: Practical takeaways, reference paper, episode transcript.

Metabolic Effects of Oxidative Stress in Development & Neurodegeneration Disease | Robert Lustig | 272
This is a free preview of a paid episode. To hear more, visit mindandmatter.substack.comWide release: January 4, 2026. Not medical advice.TOPICS DISCUSSED:* Cellular growth vs. burning: Cells alternate between growing (using glucose for building blocks in low-oxygen environments) and burning (generating ATP in mitochondria with oxygen); dysregulation leads to metabolic issues.* Key regulatory enzymes: PI3 kinase imports glucose, AMP kinase builds mitochondria, and mTOR drives cell division; their synchronization determines healthy modes, while desynchronization causes diseases.* Fructose as a dose-dependent mitochondrial toxin: High fructose intake inhibits AMP kinase, reducing mitochondrial function and diverting energy to fat storage; it is dose-dependent, like alcohol, and unnecessary in the diet.* Obesogens & endocrine disruptors: Chemicals like tributyltin (TBT) alter gene expression across generations, promoting obesity unrelated to calories; modern exposures increase reactive oxygen species (ROS), burdening cells.* Fetal & neonatal development: Maternal diet, especially high sugar or formula feeding, can cause neonatal obesity and fatty liver; breastfeeding supports proper jaw development and oxygen intake.* Brain metabolism & Alzheimer’s: The brain’s high energy needs make it vulnerable to mitochondrial issues and ROS; energy deficits from diet, stress, and toxins lead to synapse loss and inflammation, treatable via prevention.* ROS & health: Mitochondria produce ROS as a byproduct of ATP generation; excess from diet or environment causes damage, but antioxidants and lifestyle can mitigate risks.ABOUT THE GUEST: Robert Lustig, MD is a pediatric endocrinologist and Professor Emeritus at the University of California, San Francisco, with a background in neuroendocrinology and obesity research.RELATED EPISODE:* M&M 145: Epigenetics, Hormones, Endocrine Disruptors, Microplastics, Xenoestrogens, Obesogens & Obesity, Inheritance of Acquired Characteristics | Bruce Blumberg* Full audio version: [Apple] [Spotify] [Elsewhere]* Full video version: [YouTube]* Support M&M if you find value in this content.SUBSCRIBER CONTENT BELOW: Practical takeaways, reference paper, episode transcript.

Carbohydrates vs Ketosis in Exercise, Fatigue & Sports Science | Andrew Koutnik | 271
This is a free preview of a paid episode. To hear more, visit mindandmatter.substack.comWide release date: December 31, 2025. Not medical advice.TOPICS DISCUSSED:* Insulin as a powerful metabolic hormone: regulates nutrient storage across tissues, overriding others like glucagon to promote fat and glucose storage during abundance.* Glucagon & GLP-1 roles in metabolism: Glucagon mobilizes liver glucose during scarcity; GLP-1, amplified in drugs like Ozempic, suppresses hunger but originated as a diabetes treatment.* Transition to ketosis in fasting or low-carb diets: Low insulin enables fat breakdown into ketones for brain fuel, allowing survival for weeks without food, with adaptation taking about four weeks.* Hypoglycemia vs. glycogen depletion: Low blood sugar causes fatigue and irritability due to brain energy deficit, while muscle glycogen levels do not directly limit performance.* Ketogenic diets & exercise performance: Studies show no difference in endurance after adaptation, with some athletes performing better on low-carb due to enhanced fat oxidation.* High-carb diets in athletes: In one study, about 30% developed prediabetes-like fasting glucose elevations, linked to total carb intake, despite leanness and fitness.* Misconceptions in sports nutrition: Guidelines recommend 60-90g carbs/hour, but evidence shows 10g suffices to maintain blood sugar and performance, avoiding insulin spikes that impair fat use.* Individual variability in diet response: Athletes vary in optimal fuel sources; it’s possible to by athletic and lean but also metabolically unhealthy.ABOUT THE GUEST: Andrew Koutnik, PhD earned a PhD in biomedical sciences with a focus on exercise physiology and metabolic health, informed by his personal diagnosis of type 1 diabetes in childhood.RELATED EPISODE:* M&M 267 | Ketosis & BHB: Metabolic Diet Therapies, Brain Cancer & Exercise | Dominic D’Agostino* Full audio version: [Apple] [Spotify] [Elsewhere]* Full video version: [YouTube]* Support M&M if you find value in this content.SUBSCRIBER CONTENT BELOW: Practical takeaways, reference paper, episode transcript.

Stress & Psilocybin: Effects on Maternal Care & Offspring Development | Danielle Stolzenberg | 270
This is a free preview of a paid episode. To hear more, visit mindandmatter.substack.comWide release date: December 26, 2025.Topics Discussed:* Maternal care behavior in rodents: Nursing, pup retrieval, grooming, and nest-building, essential for altricial pups’ survival; conserved across mammals but varies by species.* Hormonal changes in pregnancy: Estradiol and progesterone surge then drop at birth, crossing the blood-brain barrier to enable infant attraction and care via gene expression and neuroplasticity.* Brain circuitry for parenting: Medial preoptic area acts as a central hub, coordinating motivation and sensory inputs; present in both sexes but activated differently by hormones and experience.* Stress impacts on mothers: Social stressors like male intruders dysregulate care, leading to frantic behaviors and avoidance; models human psychosocial stress linked to postpartum mood disorders.* Sex differences in pup care: Mothers groom male pups more, influencing sexual behaviors, which effects future behavior.* Psilocybin in postpartum mice: Single dose increased anxiety in mothers, showed no antidepressant effects, and transferred via milk, causing long-term anhedonia and impairments in offspring as adults.* Serotonin system development: Early exposure to serotonergic drugs like psilocybin or SSRIs alters lifelong behavior, highlighting sensitive periods in brain reorganization.Practical Takeaways:* Reduce postpartum stress through social support to enhance maternal bonding and minimize mood disorder risks.* Approach psychedelics cautiously during postpartum due to potential anxiety increases and offspring effects via milk.* Recognize hormonal shifts heighten sensitivity to infant cues, aiding natural caregiving instincts.* Monitor environmental factors like food availability or threats that could disrupt parental behaviors in high-stress scenarios.About the guest: Danielle Stolzenberg, PhD is an associate professor of psychology at the University of California, Davis, where her lab studies the neurobiology of maternal care.Reference Paper:* Study: Psilocybin during the postpartum period induces long-lasting adverse effects in both mothers and offspringRelated Episode:* M&M 180: Mother-Infant Bonding, Maternal Care & Breastfeeding, Neural Basis of Hunger & Social Behavior | Marcelo Dietrich*Not medical advice.* Full audio version: [Apple] [Spotify] [Elsewhere]* Full video version: [YouTube]* Support M&M if you find value in this content.* Episode transcript below.Full AI-generated transcript below. Beware of typos & mistranslations!

Soybean Oil: Obesity, Fatty Liver Disease, Gut Health, IBS & Colitis | Frances Sladek | 269
This is a free preview of a paid episode. To hear more, visit mindandmatter.substack.comWide release date: December 21, 2025.Topics Discussed:* Historical trends in soybean oil use: Consumption increased dramatically since the 1960s due to farming subsidies, now providing over 10% of calories for many Americans, far exceeding the 1-2% required biologically.* Soybean oil’s effects in mice: Diets with 8-10% linoleic acid cause obesity, fatty liver, and diabetes over weeks, unlike coconut oil diets; effects persist even after diet switch unless combined with fasting.* Role of HNF4 protein: This conserved liver transcription factor binds linoleic acid, regulating gene expression for metabolism; variants shift between carbohydrate and fat processing, with imbalances linked to fatty liver and cancer.* Oxylipins from linoleic acid: Conversion in liver drives obesity; mice unable to produce them resist weight gain on soybean oil, suggesting these metabolites are key culprits.* Vitamin B1 & soybean oil: Diets deplete B1 in liver and blood, contributing to obesity; supplementation with B1 analogs prevents weight gain, unlike beef tallow diets which preserve B1 levels.* Gut & microbiome impacts: Soybean oil alters gut bacteria, potentially reducing B1 production and increasing permeability, leading to inflammation; farm animals fed soybean meal pass effects to consumers.* Broader health implications: Reanalysis of old human studies questions linoleic acid’s heart benefits; focus on reducing processed foods and seed oils, while noting olive oil’s advantages from historical contexts.Practical Takeaways:* Limit processed foods and seed oils like soybean to reduce linoleic acid intake, aiming for 1-2% of calories; read labels and opt for olive oil or home-cooked meals.* Incorporate intermittent fasting, such as 12-16 hours without eating daily, to help reverse obesity effects from high-linoleic diets, based on mouse reversibility studies.* Choose grass-fed or naturally fed animal products to avoid indirect soybean oil exposure from feed, potentially preserving nutrient levels like vitamin B1.* Monitor diet when traveling or changing habits, as shifts in oil types can affect medication metabolism via liver enzymes.About the guest: Frances Sladek, PhD is a professor whose research focuses on the nuclear receptor HNF4 and the health impacts of dietary fats, particularly soybean oil.Reference Paper:* Study: Diet high in linoleic acid dysregulates the intestinal endocannabinoid system and increases susceptibility to colitis in MiceRelated Episode:* M&M 200: Dietary Fats & Seed Oils in Inflammation, Colon Cancer & Chronic Disease | Tim Yeatman & Ganesh Halade*Not medical advice.* Full audio version: [Apple] [Spotify] [Elsewhere]* Full video version: [YouTube]* Support M&M if you find value in this content.* Episode transcript below.Full AI-generated transcript below. Beware of typos & mistranslations!

Dietary Fat & Light Regulation of Circadian Biology | Louis Ptacek | 268
This is a free preview of a paid episode. To hear more, visit mindandmatter.substack.comWide release date: December 17, 2025.Topics Discussed:* Evolutionary context of circadian rhythms: All organisms have adapted to Earth’s 24-hour day for survival, with internal clocks slightly offset and adjusted by environmental cues.* Molecular clock mechanism: Involves a feedback loop where proteins turn on/off genes, lasting ~24 hours, regulated by phosphorylation and degradation for timing precision.* Genetic variations in sleep: Families with mutations in clock genes like PER2 cause extreme morning lark behavior, altering protein stability and period length by hours.* Light entrainment: Morning light shortens human clocks (average 24.2 hours) to match 24-hour days; seasonal day length changes require gradual adjustments.* Food & metabolic links: Seasonal food scarcity/abundance affects clock via glucose and fatty acids competing for protein modifications, as shown in diabetic mouse models.* Role of unsaturated fats: Paper finds MUFA/PUFA ratios in diet alter phosphorylation of clock proteins, speeding or slowing adaptation to winter/summer light cycles in mice.* Modern environmental impacts: Artificial light extends “daytime” signals, while constant food access erases seasonal patterns, contributing to obesity and diabetes risks.* Jet lag & adaptations: Sudden time shifts mimic seasonal experiments; high-sugar/fat intake may phenocopy genetic effects to aid adjustment, though not recommended for health.Practical Takeaways:* Expose yourself to morning natural light to help synchronize your internal clock and improve daily energy.* Consume main meals during daylight hours and avoid late-night eating to align with natural metabolic rhythms.* Limit evening screen time to reduce artificial blue light disrupting sleep onset.* Consider varying diet seasonally, favoring diverse, whole foods to mimic natural availability patterns for better health.About the guest: Louis Ptacek, MD is a neurologist and professor at the University of California, San Francisco. He researches inherited neurological diseases and sleep traits, including genetic variations causing extreme early rising.Reference Paper:* Study: Unsaturated fat alters clock phosphorylation to align rhythms to the season in miceRelated Episode:* M&M 237: Circadian Biology: Genetics, Behavior, Metabolism, Light, Oxygen & Melatonin | Joseph Takahashi*Not medical advice.* Full audio version: [Apple] [Spotify] [Elsewhere]* Full video version: [YouTube]* Support M&M if you find value in this content.* Episode transcript below.Full AI-generated transcript below. Beware of typos & mistranslations!

Ketosis & BHB: Metabolic Diet Therapies, Brain Cancer & Exercise | Dominic D'Agostino | 267
This is a free preview of a paid episode. To hear more, visit mindandmatter.substack.comWide release date: December 12, 2025.Topics Discussed:* Organs have different fuel preferences: brain strongly prefers glucose, heart prefers fatty acids, skeletal muscle is flexible and likes fat/ketones.* Humans evolved with high metabolic flexibility; regular ketosis was normal for ancestors, but today most people never experience it.* “Keto flu” is largely glucose withdrawal plus electrolyte/sodium loss; proper salt and hydration prevent most symptoms.* Classic medical ketogenic diet is ~90% fat (historically saturated); modern versions often use more monounsaturated fats, MCTs, and higher protein.* Saturated fat is not inherently atherogenic in the context of weight stability or caloric deficit; excess calories from any source can dysregulate metabolism.* Exogenous ketones (e.g. BHB) provide energy, reduce ROS, stabilize membranes, increase inhibitory tone (GABA), and have hormone-like signaling effects independent of diet.* Cancer cells often show Warburg effect (damaged mitochondrial respiration → heavy reliance on glycolysis); lowering glucose and raising ketones can stress cancer cells.* True keto-adaptation for athletic performance requires 6–12 weeks; after that, elite athletes can match or exceed prior high-carb performance at sub-maximal and endurance efforts.Practical Takeaways:* Therapeutic carbohydrate restriction (50–100 g/day for many people) plus occasional fasting or ketone supplements can restore metabolic flexibility with far fewer side effects than strict keto.* Prioritize whole-food fats (eggs, fatty fish, beef, olive oil, butter/lard) and minimize processed keto products loaded with seed oils.* Supplementing BHB (salts or esters) or MCT oil can ease the transition into ketosis, boost ketones without strict dieting, and may support brain and metabolic health.* Regularly check basic blood markers (glucose, lipids, electrolytes) and consider an OmegaQuant test; optimizing metabolic health is one of the strongest preventable steps against cancer, neurodegeneration, and heart disease.About the guest: Dominic D’Agostino, PhD, is an associate professor at the University of South Florida Morsani College of Medicine in the Department of Molecular Pharmacology and PhysiologyRelated Episode:* M&M 224: Cancer Biology: Metabolism, Mitochondria & Energy | Thomas SeyfriedSupplemental Ketone (BHB):* KetoCitra—Ketone body BHB with potassium, calcium & magnesium, formulated with kidney health in mind. Use code MIND20 for 20% off.*Not medical advice.* Full audio version: [Apple] [Spotify] [Elsewhere]* Full video version: [YouTube]* Support M&M if you find value in this content.* Episode transcript below.Full AI-generated transcript below. Beware of typos & mistranslations!

Seed Oils, Chronic Inflammation, Heart Health & Marijuana | Ganesh Halade | 266
This is a free preview of a paid episode. To hear more, visit mindandmatter.substack.comWide release date: December 7, 2025.Topics Discussed:* Polyunsaturated fatty acids (PUFAs): Omega-6 from seed oils like safflower and corn can convert to pro-inflammatory molecules, while omega-3s produce resolving ones; imbalance biases toward chronic inflammation.* Inflammation regulation: Acute inflammation aids healing but requires active “on” and “off” signals from lipid mediators; chronic inflammation arises from excess omega-6, delaying resolution.* Heart health & diet: High omega-6 diets worsen post-heart attack outcomes in mice by elevating pro-inflammatory lipids.* Evolution & historical context: PUFAs are essential but naturally balanced in pre-industrial diets; modern processing skews ratios, contributing to diseases, as shown in early rat experiments needing minimal fats for survival.* Aging & lifestyle factors: Excess omega-6 exacerbates inflammation in older mice; sleep, exercise, and balanced fats are crucial for metabolic health and enzyme function in processing lipids.* Cannabis & omega-6 interaction: In mice on high omega-6 diets, CBD-rich cannabis smoke reaches the heart quickly, suppresses immune response, and weakens cardiac strain, unlike in balanced-diet controls.* Genetic models: FAT-1 mice converting omega-6 to omega-3 show better healing and neuroprotection; FAT-2 mice doing the opposite exhibit liver fibrosis and metabolic issues, highlighting omega-6 excess harms.Practical Takeaways:* Balance omega-6 and omega-3 intake by reducing seed oils in processed foods and increasing sources like fish or algae to support inflammation resolution and heart health.* Monitor and adjust fat intake with age, as older individuals are more sensitive to omega-6 excess leading to immune dysregulation.* Avoid combining high omega-6 diets with smoking, as it may impair immune and cardiac responses based on animal data.About the guest: Ganesh Halade, PhD is a cardiovascular scientist and associate professor at the University of South Florida Morsani College of Medicine.Reference Paper: * Paper: Cannabis cigarette smoking disrupts mice multi-organ bioactive lipid metabolism and inflammation-resolution signaling in an obesogenic settingRelated Episode:* M&M 200: Seed Oils, Inflammation, Colon Cancer & Chronic Disease | Tim Yeatman & Ganesh Halade *Not medical advice.* Full audio version: [Apple] [Spotify] [Elsewhere]* Full video version: [YouTube]* Support M&M if you find value in this content.* Episode transcript below.Episode Chapters:00:03:38 Guest Introduction & Background 00:04:42 Polyunsaturated Fatty Acids Overview 00:08:31 Acute vs Chronic Inflammation 00:12:01 Inflammation Turn-On & Turn-Off Signals 00:17:59 Omega-6 & Omega-3 Imbalance in Modern Diet 00:23:39 Essential Fatty Acids & Natural Balance 00:28:09 Seed Oils & Heart Health Controversy 00:37:27 High Omega-6 Effects on Heart Health 00:45:32 Aging & Omega-6 Sensitivity 00:53:55 Healing Faster with Balanced Ratio 00:59:36 Cannabis Smoking & High Omega-6 Diet 01:11:19 Immune Dysregulation from Cannabis & Omega-6 01:15:00 Ongoing Heart Failure Research 01:21:26 FAT-1 & FAT-2 Mice Insights 01:28:10 Lifestyle Medicine & Final ThoughtsFull AI-generated transcript below. Beware of typos & mistranslations!

Evaluating Science: Clinical Trials, Epidemiology, Preclinical Studies & Mendelian Randomization | George Davey Smith | 265
This is a free preview of a paid episode. To hear more, visit mindandmatter.substack.comWide release date: December 2, 2025.Topics Discussed:* Epidemiology basics: Studies disease influences using observational designs like case-control and prospective cohorts, plus trials, to identify patterns and test hypotheses.* Hierarchy of evidence critique: Rejects rigid pyramids favoring RCTs, as all studies can be biased; advocates triangulation integrating varied data types for robust conclusions.* RCT strengths & weaknesses: Randomization balances confounders, but issues like poor blinding, attrition, or subversion can undermine results; large samples may yield spurious precision if biased.* Confounding & reverse causation: Examples include yellow fingers and lung cancer (both from smoking) or early atherosclerosis inflating CRP-disease links; hard to fully control statistically.* Nutrition epidemiology pitfalls: Observational studies often overstate benefits (e.g., vitamin E for heart disease), leading to failed trials; incentives favor new findings over revisiting errors.* Mendelian randomization: Uses genetic variants as proxies for exposures (e.g., ALDH2 for alcohol metabolism) to mimic randomization; reveals no heart benefits from alcohol, unlike observational data.* Negative controls for validation: Tests implausible outcomes (e.g., smoking and murder) or exposures (e.g., paternal smoking in pregnancy) to check for confounding artifacts.* Evidence triangulation: Combines diverse studies with different biases (e.g., cross-cultural comparisons) for causality; applied to dismiss HDL-raising drugs despite initial promise.Practical Takeaways:* Scrutinize health claims by checking for negative controls or variety in evidence sources to avoid mistaking correlation for causation.* For personal decisions like alcohol intake, consider genetic studies showing risks at all levels, and aim for moderation or abstinence based on overall evidence.* When evaluating supplements or diets, prioritize trials over observational data, and question media hype that ignores confounding factors.* Use symmetrical analysis in reading studies: Treat exposures and confounders equally to assess true effects.About the guest: Dr. George Davey Smith, MD, DSc is a professor of clinical epidemiology at the University of Bristol and director of the MRC Integrative Epidemiology Unit, where he focuses on causal inference in health.Reference Paper: * Paper: Evidence triangulation in health researchRelated Episode:* M&M 212: How Science Really Works: Meta-Research, Publishing, Reproducibility, Peer Review, Funding | John Ioannidis*Not medical advice.* Full audio version: [Apple] [Spotify] [Elsewhere]* Full video version: [YouTube]* Support M&M if you find value in this content.* Episode transcript below.Episode Chapters:00:00:00 Intro 00:06:01 Hierarchy of Evidence 00:12:54 Sample Size & Precision 00:18:41 Vitamin E Supplements 00:25:05 Nutrition Epidemiology Pitfalls 00:32:01 Preclinical vs Clinical 00:38:04 Negative Controls 00:45:16 Negative Control Exposures 00:52:21 Alcohol Consumption Effects 00:59:00 Mendelian Randomization Example 01:05:16 MR Limitations & Pleiotropy 01:11:20 HDL Cholesterol Myths 01:18:21 Evidence Triangulation 01:23:36 Final Thoughts & ResourcesFull AI-generated transcript below. Beware of typos & mistranslations!

Energy Metabolism & Information Processing in the Brain | Luis Felipe Barros | 264
This is a free preview of a paid episode. To hear more, visit mindandmatter.substack.comWide release date: November 26, 2025.Topics Discussed:* Brain energy efficiency: Brains are much more energy-efficient than computers for similar processing, relying on adaptive metabolic strategies evolved under energy scarcity.* Metabolism vs. information processing: Core metabolites like glutamate bridge basic cellular energy production and neural signaling.* Lactate as a signal: Produced during exercise, lactate diffuses from muscles to brain, modulating neuronal excitability and providing neuroprotection.* BHB in ketosis: During fasting or ketogenic diets, beta-hydroxybutyrate displaces glucose as fuel, enhances antioxidant defenses, and activates protective potassium channels in neurons.* Adenosine and sleep: Accumulates from ATP breakdown during wakefulness, triggering sleep and locally inhibiting overactive neural networks for energy conservation.* Glial cells’ role: Astrocytes and oligodendrocytes handle sophisticated metabolism, release signaling molecules like lactate, and modulate synapses, and influence diseases like Alzheimer’s.Practical Takeaways:* Regular exercise elevates lactate, which signals the brain to adapt metabolism and may enhance neuroprotection against stress.* Intermittent fasting or ketogenic diets can induce ketosis with BHB, potentially boosting brain antioxidant defenses and preconditioning against metabolic stress.* Prioritizing sleep helps clear adenosine buildup, restoring energy balance and supporting long-term neural health.About the guest: Dr. Luis Felipe Barros, MD, PhD is a Chilean neurobiologist and professor at the Universidad de Valparaíso, where he leads a lab studying brain metabolism from glucose transport to mitochondrial function.Reference Paper: * Paper: Scale-spanning crosstalk between metabolism and information processingRelated Episode:* M&M 255 | Unlocking Energy: How Nutrition & Drugs Impact Your Mitochondria | Chris Masterjohn*Not medical advice.* Full audio version: [Apple] [Spotify] [Elsewhere]* Full video version: [YouTube]* Support M&M if you find value in this content.* Episode transcript below.Episode Chapters:00:00:00 Intro 00:06:00 Brain Efficiency & Computers 00:12:00 Metaphors for Brain Function 00:18:00 Core Metabolites & Signaling 00:24:00 Lactate in Exercise & Protection 00:30:00 Body-Brain Integration & Sensors 00:36:00 BHB, Ketosis & Fasting 00:42:00 Adenosine & Sleep Cycles 00:48:00 Neuroprotective Core Metabolites 00:54:00 Scale-Spanning Coordination 01:00:00 Neural Energy Demands 01:05:00 Expensive Tissue Hypothesis 01:08:00 Glial Cells & Astrocytes 01:13:00 Mitochondria in Cells 01:18:00 Future of Metabolism ResearchFull AI-generated transcript below. Beware of typos & mistranslations!

Protein Restriction & Liver Hormones: Appetite, Brain, Behavior | Chris Morrison | 251
This is a free preview of a paid episode. To hear more, visit mindandmatter.substack.comWide release date: September 12, 2025Episode Summary: Dr. Christopher Morrison talks about how animals sense and prioritize nutrients like protein, discussing defense mechanisms for essentials such as oxygen, water, sodium, and energy; the brain's role in detecting protein deprivation via signals like FGF21; trade-offs between growth, reproduction, and longevity under protein restriction; and reconciling high-protein diets for satiety and muscle maintenance with low-protein benefits for metabolic health and lifespan extension.About the guest: Christopher Morrison, PhD is a professor and researcher at the Pennington Biomedical Research Center in Baton Rouge, Louisiana, where he has worked for over 22 years focusing on nutrition, metabolism, and chronic diseases like obesity and diabetes.Discussion Points:* The body prioritizes nutrients hierarchically: oxygen and water first, then sodium, energy, and protein, with weaker defenses for carbs or fats.* Animals develop specific appetites for deprived nutrients, like salt or protein, often through post-ingestive learning rather than just taste.* Protein restriction (e.g., 5% vs. 20% in diets) increases food intake and energy expenditure in mice to maintain protein levels, even at the cost of extra calories.* FGF21, a liver hormone, signals protein deprivation to the brain (via NTS region), driving protein-seeking behavior and metabolic changes; it's essential for low-protein responses.* Protein restriction extends lifespan in lab animals by suppressing growth signals like IGF-1 and mTOR, but may impair immunity or wound healing in real-world conditions.* High protein aids satiety, weight loss, and muscle building, but overconsumption may shorten lifespan; optimal intake depends on age, activity, and goals (e.g., not for pregnant or elderly).* No one-size-fits-all for protein: mild restriction may benefit middle-aged sedentary people for health, while athletes need more; balance avoids excesses.Related content:* M&M 106: Diet, Macronutrients, Micronutrients, Taste, Whole vs. Processed Food, Obesity & Weight Loss, Comparative Biology of Feeding Behavior | Stephen Simpson & David Raubenheimer*Not medical advice.* Full audio version: [Apple] [Spotify] [Elsewhere]* Full video version: [YouTube]* Support M&M if you find value in this content.* Episode transcript below.Episode Chapters:00:00:00 Intro00:06:10 Nutrient Detection & Adequacy 00:11:11 Fluid & Sodium Defenses 00:16:20 Protein vs Other Macronutrients 00:21:21 Post-Ingestive Learning & Flavors 00:26:34 Hyperphagia on Low Protein 00:31:10 Restriction Effects on Growth 00:36:33 Longevity & Restriction Trade-Offs 00:41:47 Behavioral Changes & Choices 00:46:44 Preference in Choice Experiments 00:53:44 Innate vs Learned Appetite 00:58:39 Protein Digestion & Signals 01:04:40 FGF21 in Fasting & Restriction 01:10:40 FGF21 Knockouts & Relevance 01:15:20 Brain Receptors & Sites 01:20:49 Growth & Longevity Trade-Offs 01:25:04 High Protein Satiety & Muscle 01:30:33 FGF21 Scaling & Future Work 01:35:39 Final Thoughts & RecommendationsFull AI-generated transcript below. Beware of typos & mistranslations!

Cognition, Form, Regeneration & Metaphysics: Does Biology Arise From Math? | Michael Levin | 250
This is a free preview of a paid episode. To hear more, visit mindandmatter.substack.comWide release date: September 7, 2025Episode Summary: Dr. Michael Levin talks about cognition manifesting at scales beyond brains, including in cells and tissues via bioelectric networks; analog vs. digital coding in biology; how bioelectric patterns guide development and regeneration (e.g., in planarians); creation of novel life forms like xenobots and anthrobots; philosophical ideas on a "platonic space" of mathematical patterns influencing biology; evolutionary trade-offs in regeneration and implications for regenerative medicine.About the guest: Michael Levin, PhD is a biologist and computer scientist who directs the Allen Discovery Center at Tufts University, focusing on bioelectricity, regeneration, and cognition in non-neural systems.Discussion Points:* Cognition isn't limited to brains; tools from neuroscience reveal learning, memory, and goal-directed behavior in cells and tissues.* Bioelectricity acts as "cognitive glue," enabling collective intelligence in non-neural systems, predating multicellular life.* Analog coding in development uses slow voltage patterns across cells, contrasting fast digital spikes in neurons.* Planarians regenerate perfectly due to bioelectric "memories"; altering patterns creates stable two-headed worms without genetic changes.* Regeneration trade-offs: Mammals prioritize quick healing over full regrowth due to infection risks and load-bearing needs.* Xenobots (from frog cells) and anthropots (from human cells) self-assemble, replicate, and heal tissues, revealing untapped cellular potentials.* Patterns in biology may stem from a "platonic space" of mathematical truths, not just evolution or physics.* Neuroscience studies mind scaling, not just neurons; diverse intelligences could exist in non-cellular systems.Related content:* M&M 95: Purpose, Value, Evolution, Consciousness, Sentience, Life & Emergence of Mind From Matter | Terrence Deacon*Not medical advice.* Full audio version: [Apple] [Spotify] [Elsewhere]* Full video version: [YouTube]* Support M&M if you find value in this content.* Episode transcript below.Episode Chapters:00:00:00 Intro00:04:52 Cognition Definition 00:09:20 Development Timescales 00:12:01 Analog Signaling 00:15:12 Network Properties 00:18:03 Goals in Morphogenesis 00:21:40 Regeneration Mechanisms 00:25:41 Planarian Patterns 00:31:43 Regenerative Trade-offs 00:38:20 Pattern Sources 00:44:21 Xenobots 00:49:26 Anthropots 00:56:28 Platonic Space 01:02:47 Mind ScalingFull AI-generated transcript below. Beware of typos & mistranslations!

Fructose, Microglia, Anxiety & Brain Development | Justin Perry | 249
This is a free preview of a paid episode. To hear more, visit mindandmatter.substack.comWide release date: September 3, 2025Episode Summary: Dr. Justin Perry talks about the body's constant cellular turnover—about 3 million cells die per second in adults (double in children and women)—handled by phagocytes like macrophages that engulf and digest debris to prevent diseases like lupus. They explore phagocytosis steps, macrophage adaptations in tissues like the brain (microglia), and how high fructose intake impairs microglial function in developing mice, leading to uncleared brain cells and anxiety-like behaviors, with implications for human neurodevelopmental disorders amid rising fructose consumption.About the guest: Justin Perry, PhD is an immunologist and clinical psychologist who leads a lab at Memorial Sloan Kettering Cancer Center focusing on how the body clears dead cells and debris to maintain homeostasis.Discussion Points:* The body turns over 1-2% of its 30 trillion cells daily, mostly blood cells, but neurons in kids and endometrium in women turnover at ~2x this rate* Phagocytosis involves "find me," "eat me," and digestion signals; failures can cause autoimmunity.* Microglia are brain macrophages that uptake fructose via GLUT5 transporter.* Early high fructose exposure (comparable to one soda daily) impairs the pruning of synapses and dead neurons.* In mice, prenatal or postnatal fructose causes phagocytosis deficits in the prefrontal cortex, leading to heightened fear responses and poor fear extinction, mimicking anxiety disorders.* Fructose correlates with rising neurodevelopmental issues like autism and anxiety; it's passed via breast milk, and liquid forms (e.g., sodas) overwhelm metabolic shields more than solid fruits.* Macrophages may hold keys to diseases from atherosclerosis to cancer; deleting GLUT5 in microglia reverses fructose's effects, hinting at evolutionary roles in aging or low-oxygen states.Related content:* M&M 215: Cancer Metabolism: Sugar, Fructose, Lipids & Fasting | Gary Patti* Article | Dietary Fructose & Metabolic Health: An Evolutionary PerspectiveReference Paper:* Study | Early life high fructose impairs microglial phagocytosis and neurodevelopment*Not medical advice.* Full audio version: [Apple] [Spotify] [Elsewhere]* Full video version: [YouTube]* Support M&M if you find value in this content.* Episode transcript below.Episode Chapters:00:00:00 IntroFull AI-generated transcript below. Beware of typos & mistranslations!

Cholesterol: Immune Benefits, Heart Health, Statins & Research Malpractice | Uffe Ravnskov | 247
This is a free preview of a paid episode. To hear more, visit mindandmatter.substack.comWide release date: August 25, 2025Episode Summary: Dr. Uffe Ravnskov talks about his decades-long career challenging the idea that high cholesterol causes heart disease, discussing LDL's protective role in the immune system by binding to bacteria, the harms and biases in statin research influenced by pharmaceutical companies, evidence that high cholesterol benefits the elderly and reduces infection/cancer risks, and how mental stress or infections elevate cholesterol as a response rather than a cause.About the guest: Uffe Ravnskov, MD, PhD is a physician and independent researcher who earned his MD from the University of Copenhagen in 1961 and a PhD in nephrology. He has worked in various clinics in Sweden since the 1960s, focusing his research on challenging the cholesterol hypothesis in heart disease. Now 91, he has published over 200 papers, authored books like "The Cholesterol Myths.”Discussion Points:* LDL cholesterol helps the immune system by sticking to bacteria, clumping them for removal; low LDL increases infection risk.* Animal studies show injecting LDL protects against lethal infections, while historical data links severe infections to worse atherosclerosis.* Elderly people with high cholesterol live longer; low cholesterol raises mortality risk more than high levels.* Familial hypercholesterolemia (FH) doesn't cause early death via cholesterol alone—co-inherited coagulation factors are the issue, and FH patients often have lower infection rates.* Statins lower LDL but increase infection risk, cause muscle weakness/brain issues (often blamed on aging), and show no clear benefit in unbiased meta-analyses.* Research biases include cherry-picking studies, exaggerating benefits via relative (not absolute) risk, and pharma funding suppressing critical views.* Mental stress can raise cholesterol by 10-50% in 30 minutes, often misread as a heart disease cause rather than an effect.* Saturated fat and high cholesterol aren't proven harmful; Ancel Keys' claims ignored contradictory evidence.* Stopping statins often reverses side effects quickly, improving quality of life.Related episode:* M&M 244: Seed Oils & Heart Disease: Oxidized LDL, Cholesterol, Fat & Cardiology | Tucker GoodrichReference Paper:* LDL-C does not cause cardiovascular disease: a comprehensive review of the current literature*Not medical advice.* Full audio version: [Apple] [Spotify] [Elsewhere]* Full video version: [YouTube]* Support M&M if you find value in this content.* Episode transcript below.Episode Chapters:00:00:00 Intro00:01:28 Guest Background & Career 00:05:15 Cholesterol & Immune Function 00:10:14 LDL Role in Infections 00:15:10 Statins & Industry Influence 00:20:26 Research Biases & Relative Risk 00:25:08 Pushback & Publication Challenges 00:30:13 FH & Coagulation Factors 00:35:02 Stress & Cholesterol Response 00:40:03 Mortality & Elderly Benefits 00:45:32 Statin Side Effects & Recommendations 00:50:30 Scandal in Medicine & OutroFull AI-generated transcript below. Beware of typos & mistranslations!
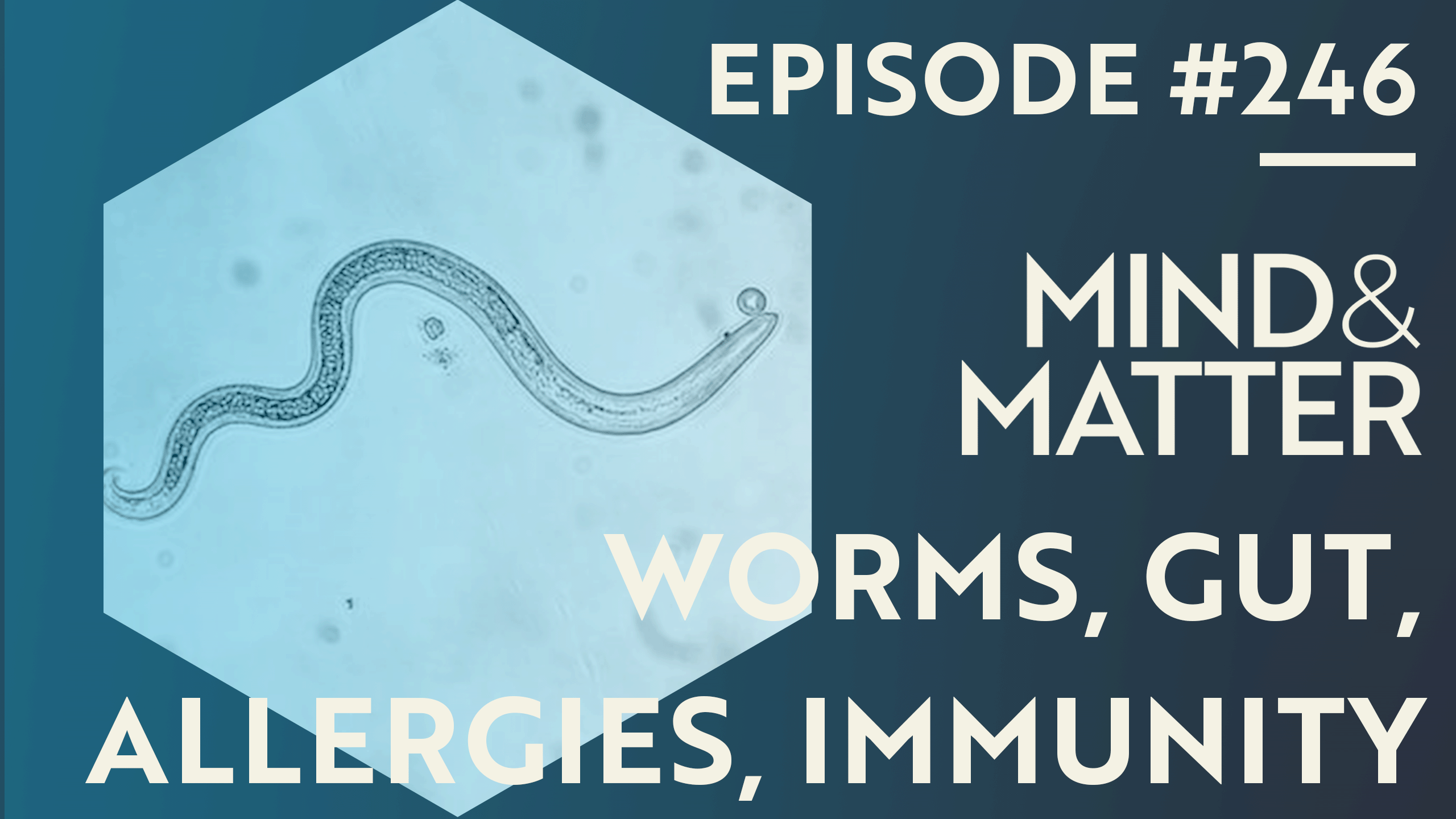
Appendix, Gut Worms, Allergies & Autoimmunity | William Parker | 246
This is a free preview of a paid episode. To hear more, visit mindandmatter.substack.comWide release date: August 18, 2025Episode Summary: Dr. William Parker discusses gut anatomy, the appendix's role in harboring beneficial bacterial biofilms and immune tissue, and how modern hygiene depletes helminths (intestinal worms), causing immune overreactions like allergies, autoimmunity, and psychiatric conditions. He explores helminth self-therapy for treating relapsing multiple sclerosis, depression, and allergies; challenges in clinical trials due to patent issues; and why COVID-19 was milder in low-income, helminth-rich regions.About the guest: William Parker, PhD conducted research at Duke University for over 27 years on immunology, appendicitis, and the hygiene hypothesis. He now serves as a visiting scholar at the University of North Carolina at Chapel Hill, leading efforts on biome reconstitution via helminths.Discussion Points:* Appendix is not vestigial; it concentrates immune tissue and biofilms to cultivate good gut bacteria, preventing pathogens via mucus and IgA antibodies.* Hygiene hypothesis: Soap, toilets, and clean water reduce helminths/protozoa, leading to untrained, hyperactive immunity and rising allergies/autoimmunity since the 1800s.* Helminths (worms) stimulate immune "exercise," training immunity; biohackers use hookworms (cheap, skin-entry), porcine whipworms, or rat tapeworms orally for relief from allergies, MS flares, depression/anxiety.* Effects are temporary; need ongoing exposure (e.g., replenish every 6 months); immigrants from helminth-rich areas develop Western diseases within a few years.* COVID-19: Hyper-immunity caused severe reactions in hygienic West, but helminth presence in low-income Africa/Asia prevented cytokine storms, leading to empty clinics.* Therapy barriers: Non-patentable organisms require $100M+ trials; push for open-source, government-funded biome restoration over crude immunosuppressants.Related episode:* M&M 144: Inflammation, Innate Immunity, Allergies & Allergens, Immune System Evolution, Fasting & Metabolism | Clare Bryant*Not medical advice.* Full audio version: [Apple] [Spotify] [Elsewhere]* Full video version: [YouTube]* Support M&M if you find value in this content.* Episode transcript below.Episode Chapters:00:00:00 Intro 00:03:24 Gut Anatomy & Appendix 0:09:18 Appendicitis & Inflammation 00:14:46 Biofilms & Immune Support 00:19:48 Hygiene Hypothesis History 00:25:24 Appendicitis Emergence 00:30:13 Appendix Evolution 00:36:21 Natural Biome Components 00:42:22 Worms & Autoimmunity 00:47:03 Biohacking with Helminths 00:52:24 Worm Administration Methods 00:57:23 Treated Conditions 01:03:08 Clinical Trials & Challenges 01:08:18 Covid-19 Morbidity Differences 01:13:23 Rat Immune Comparisons 01:15:52 Final Thoughts & FutureFull AI-generated transcript below. Beware of typos & mistranslations!

Cannabinoid System: Metabolism, Evolution & Energy Storage | Giovanni Marsicano | 245
This is a free preview of a paid episode. To hear more, visit mindandmatter.substack.comWide release date: August 13, 2025Episode Summary: Dr. Giovanni Marsicano is a neuroscientist based in Bordeaux, France, where he leads a research group at INSERM focusing on the endocannabinoid system.About the guest: Giovanni Marsicano, PhD discusses the endocannabinoid system, starting with its core components like CB1 receptors and lipid-based molecules such as anandamide and 2-AG, derived from omega-6 fatty acids; he explains its cellular signaling, evolutionary role in energy storage for uncertain futures (exostasis vs. homeostasis), and effects across tissues like the liver, gut, and brain, including motivation, appetite, pain relief, and anxiety regulation, while touching on biphasic effects of cannabinoids.Discussion Points:* The endocannabinoid system acts as an "exostatic" regulator, promoting energy accumulation for future needs by enhancing food palatability, nutrient absorption, and fat storage, unlike "endostatic" systems that address immediate hunger.* CB1 receptors appear in vertebrates with adipose tissue, suggesting an evolutionary link to storing fat for survival in unstable environments.* Endocannabinoids are lipids from omega-6 fats; high intake boosts their levels, potentially fueling obesity by creating a self-perpetuating cycle of overeating.* Activation of CB1 can have biphasic effects (e.g., low doses reduce anxiety, high doses increase it), due to receptors on different cell types like excitatory vs. inhibitory neurons.* Pregnenolone, a steroid precursor, acts as a natural CB1 inhibitor to prevent excessive activation, blocking harmful effects like psychosis from high THC doses.* The system influences motivation beyond food, including sex and even human activities like sports or storytelling, by rewarding actions for potential future benefits.* In the brain, CB1 on mitochondria and astrocytes modulates energy use, olfaction, and social stress transmission, with implications for disorders like Alzheimer's.Reference Paper:* The CB1 Receptor as the Cornerstone of ExostasisRelated episode:* M&M 123: Endocannabinoids, Stress, Exercise, Cortisol, Anxiety, Cannabis & Effects of Marijuana on Brain Development | Matthew Hill*Not medical advice.* Full audio version: [Apple] [Spotify] [Elsewhere]* Full video version: [YouTube]* Support M&M if you find value in this content.* Episode transcript below.Episode Chapters:00:00:00 Intro00:05:01 CB1 Receptor Overview00:10:58 Endocannabinoids & Lipids 00:16:00 CB1 as GPCR 00:21:04 Mitochondrial Signaling 00:26:09 Biphasic Effects 00:31:13 Homeostasis & Energy 00:37:40 Unstable Environments 00:43:59 ECS as Exostatic 00:50:13 Cue-Induced Feeding 00:55:00 Gut & Glucose Effects 01:00:32 Omega-6 Link 01:07:02 Pregnenolone Inhibitor 01:12:05 Modulation Mechanisms 01:17:03 Clinical Applications 01:22:15 Exostatic Coordination 01:26:19 Rewards Beyond Food 01:31:39 Cultural Motivations 01:36:43 Current Lab Work 01:42:12 Final ThoughtsFull AI-generated transcript below. Beware of typos & mistranslations!

Evolution & Variation in Human Diet, Energy Expenditure & Metabolism | Herman Pontzer | 243
This is a free preview of a paid episode. To hear more, visit mindandmatter.substack.comWide release date: August 1, 2025Episode Summary: Anthropologist Dr. Herman Pontzer discusses human evolution and metabolism, comparing humans to primates like chimps and gorillas to explain our higher energy use, bigger brains, and longer lives despite trade-offs in reproduction and activity; they discuss dietary shifts from plant-based to hunting-gathering, metabolic adaptations, and modern issues like obesity, where exercise aids health but diet drives weight loss, emphasizing ultra-processed foods' role in overeating and the promise of new drugs like GLP-1 agonists.About the guest: Herman Pontzer, PhD is a professor of evolutionary anthropology and global health at Duke University. He is the author of books like "Burn" and "Adaptable," which explore how bodies adapt to diets, activity, and environments.Discussion Points:* Humans burn 20% more daily energy than other primates (controlling for body size), enabling big brains, more babies, and longer lives, but requiring efficient food strategies like hunting and gathering.* Unlike apes, humans evolved smaller guts, higher body fat (15-30% vs. apes' * Hunter-gatherers like the Hadza are far more active than sedentary Westerners but burn similar calories, as bodies adapt by reducing basal metabolism, inflammation, and hormones.* Obesity stems more from increased calorie intake via ultra-processed foods than it does from reduced activity; exercise boosts health but rarely causes major weight loss on its own.* Early humans likely scavenged rancid meat, evidenced by low stomach pH similar to vultures, aiding digestion of risky foods.* Ketosis isn't unique to high-meat diets; even Inuit on low-carb diets resist it, and other carnivores don't stay in ketosis constantly.* Across global populations, richer countries have higher BMI and slightly higher total energy expenditure due to larger bodies, but basal rates drop with lower pathogen loads.* For weight loss, Dr. Pontzer points to diets high in fiber/protein for satiety; intermittent fasting works by cutting calories, not magic; GLP-1 drugs mimic hormones to curb hunger, although we stay mindful of potential long-term effects.Related episode:* M&M 160: Diet, Hunting, Culture and Evolution of Paleolithic Humans & Hunter Gatherers | Eugene Morin*Not medical advice.* Full audio version: [Apple] [Spotify] [Elsewhere]* Full video version: [YouTube]* Support M&M if you find value in this content.* Episode transcript below.Episode Chapters:00:00:00 Intro 00:04:07 Life History & Energy Expenditure 00:09:26 Gut-Brain Trade-Off 00:15:13 Measuring Metabolism 00:20:38 Metabolic Scaling Laws 00:25:34 Evolutionary Energy Boost 00:30:29 Activity Levels Compared 00:35:13 Genus Homo Origins 00:41:18 Water Conservation Adaptations 00:46:07 Scavenging & Rancid Meat 00:52:00 Metabolic Flexibility 00:58:24 Ketosis in Diets 01:03:18 Obesity Myths 01:08:10 Activity Adaptations 01:13:43 Diet & Obesity 01:19:05 Ultra-Processed Foods 01:24:55 Global Energy Study 01:31:15 Development Effects 01:35:33 Weight Loss Strategies 01:41:23 GLP-1 Drugs 01:46:11 Book OverviewFull AI-generated transcript below. Beware of typos & mistranslations!

Sunlight: Cosmology, Energy Metabolism & the Physics of Life | Robert Fosbury | 242
This is a free preview of a paid episode. To hear more, visit mindandmatter.substack.comWide release date: July 27, 2025Episode Summary: Astrophysicist Dr. Robert Fosbury discusses the sun's characteristics as a star, its analogies to living systems via entropy and complexity, and Erwin Schrödinger's insights on life as order-maintaining entities; he explores how near-infrared (NIR) light from the sun penetrates bodies to enhance mitochondrial function and metabolism, critiques modern artificial lighting's health impacts like mitochondrial dysfunction leading to diseases, and advocates returning to natural light environments for better wellness, drawing connections from cosmology to everyday architecture and lifestyle.About the guest: Robert Fosbury, PhD is a is a retired astrophysicist. He spent his career at the European Space Agency, working on Hubble and JWST projects, and now pursues interdisciplinary research linking stellar phenomena to biological processes like light's impact on vision and metabolism.Discussion Points:* Stars like the sun maintain low-entropy states by exporting entropy as light, mirroring how life ingests low-entropy food to sustain order and homeostasis.* The universe's complexity peaks midway in entropy increase, with stars producing elements that enable biological complexity, evolving toward cognition.* Near-infrared light, peaking in solar output at ~1.6 microns due to atmospheric physics, catalyzes mitochondrial ATP production by facilitating electron transport, not via photosynthesis but photo-metabolism.* Modern LEDs and windows block near-infrared, contributing to mitochondrial dysfunction, obesity, diabetes, and aging; historical thermal lights like incandescents provided beneficial infrared.* Outdoor environments, especially under trees, flood bodies with reflected near-infrared for health, while blue skies act as cold sinks boosting thermodynamic efficiency.* Eyes are mitochondria-rich, vulnerable to poor light; therapies using near-infrared slow macular degeneration by improving energy production.* Ultra-processed foods are "high-entropy" with no structural order, akin to waste, reducing nutritional value for maintaining bodily order.* Practical fixes: Use low-voltage incandescents indoors, prioritize outdoor time, design buildings with infrared-transmitting glass, and light people, not spaces, for energy savings and health.Related episode:* M&M 146: Photobiology, Sunlight, Firelight, Incandescent Bulbs vs. LEDs, Mitochondria, Melatonin, Sunscreen & the Optics of the Body | Scott Zimmerman*Not medical advice.* Full audio version: [Apple] [Spotify] [Elsewhere]* Full video version: [YouTube] * Support M&M if you find value in this content.* Episode transcript below.Episode Chapters:* 00:00:00 Intro* 00:02:50 Background & Collaboration* 00:04:57 Reindeer Vision* 00:09:19 Sun as Star* 00:15:01 Stars & Entropy* 00:17:30 Schrodinger's Book* 00:21:05 Sun's Lifespan* 00:26:39 Solar Output & Life* 00:33:43 Schrodinger's Insights* 00:39:40 Thermodynamics & Life* 00:44:31 Star-Life Analogy* 00:48:37 Complexity & Entropy* 00:53:01 Infrared in Nature* 00:59:19 Entropy in Food* 01:13:43 Peak Photon Flux* 01:19:00 Evolution & Light* 01:24:08 Light Penetration* 01:30:34 Effects on Mitochondria* 01:37:47 Modern Light Environment* 01:43:14 Space & Dysfunction* 01:49:05 Retina & Health* 01:56:27 Remedies & Outdoors* 02:03:16 Architecture & Design* 02:08:02 Thermodynamic EfficiencyFull AI-generated transcript below. Beware of typos & mistranslations!

Synapse Energetics & Fat Metabolism in the Brain | Timothy Ryan | 241
This is a free preview of a paid episode. To hear more, visit mindandmatter.substack.comWide release date: July 23, 2025Episode Summary: Dr. Timothy Ryan talks about the high energy costs of synapses, the role of mitochondria and glycolysis, and challenge the long-held view that the brain relies solely on glucose by discussing new evidence that neurons burn fats from lipid droplets for fuel, especially during activity. The talk touches on metabolic flexibility, links to epilepsy treatments like ketogenic diets, neurodegenerative diseases, and future research on brain energy sources.About the guest: Timothy Ryan, PhD is a professor of biochemistry at Cornell University. His research focuses on the molecular mechanisms of synaptic transmission, particularly vesicle recycling and the bioenergetics that power neural communication. Discussion Points:* Synapses are tiny structures with ~100 vesicles per site (on average), converting electrical signals to chemical ones.* Brain tissue is energy-hungry due to trillions of synapses (in humans), relying on local mitochondria (present in only ~half of synapses) and glycolysis.* Vesicles use proton pumps and transporters to concentrate neurotransmitters, requiring ATP to maintain gradients.* Neurons are metabolically fragile; cutting fuel supply quickly impairs synapses.* Dogma held brains don't burn fats, but new work shows neurons form lipid droplets (fat stores) that are invisible because constantly used for energy via beta-oxidation.* Silencing neurons builds lipid droplets like resting muscle; activity speeds their breakdown, indicating demand-driven fat use.* Inhibiting neuron-specific fat-processing enzymes accumulates droplets and induces torpor (hibernation-like state) in animals, signaling metabolic stress.* Ketogenic diets aid epilepsy by shifting to ketones; fats may explain this, with potential ties to aging and neurodegeneration like Alzheimer's.* Brain may be a "hybrid" fuel user (glucose + fats), with open questions on fat sources and roles in cognition or disease protection.Related episode:* M&M 158: Ketosis & Ketogenic Diet: Brain & Mental Health, Metabolism, Diet & Exercise, Cancer, Diabetes | Dominic D'Agostino*Not medical advice.* Full audio version: [Apple] [Spotify] [Elsewhere]* Full video version: [YouTube] * Support M&M if you find value in this content.* Episode transcript below.Episode Chapters:00:00:00 Intro00:04:57 Synaptic Vesicle Recycling 00:09:19 Proton Pumps & Neurotransmitter Loading 00:15:09 Mitochondria at Synapses 00:20:09 Synaptic Machinery Energy Use 00:26:01 Glucose as Brain Fuel 00:32:56 Lipid Droplets & Fat Storage 00:39:46 Lipid Synthesis in ER 00:45:10 Limitations of Old Experiments 00:52:25 Disease-Linked Lipases 00:57:29 Lipase Inhibition Experiments 01:03:50 Activity-Dependent Fat Metabolism 01:06:02 Inducing Torpor State 01:13:16 Future Directions 01:19:41 Brain Energy & DiseaseFull AI-generated transcript below. Beware of typos & mistranslations!

Microbiome as a Modifiable Organ System | Eugene Chang | 240
This is a free preview of a paid episode. To hear more, visit mindandmatter.substack.comWide release date: July 18, 2025Episode Summary: Dr. Eugene Chang talks about the microbiome’s role as a vital organ, the impacts of antibiotics and Western diets on microbial health, and strategies for restoring a damaged microbiome through diet and fecal microbial transplants. They delve into microbiome dysbiosis, its links to modern diseases, and Chang’s research on personalized microbiome interventions.About the guest: Eugene Chang, MD is a physician-scientist and Professor of Medicine at the University of Chicago, specializing in gastroenterology. His research focuses on the gut microbiome as a vital organ influencing metabolic and immune health.Discussion Points:* The gut microbiome is a vital organ, acquired early in life, that supports metabolic and immune functions, but can be disrupted by antibiotics, leading to diseases like C. difficile colitis.* Western diets, high in saturated fats and low in fiber, contribute to microbiome dysbiosis, linked to modern conditions like inflammatory bowel diseases, obesity, and allergies.* Microbiome health is better assessed by functional markers (e.g., short-chain fatty acid production) than taxonomic diversity, as diversity varies widely among healthy individuals.* A patient with severe food intolerance due to antibiotic-induced microbiome damage was treated over 50 weeks with a tailored diet, restoring healthy microbiome function.* Different dietary fibers (e.g., beans vs. seaweed) are metabolized at varying rates, affecting gut health; fermented foods like kefir can bypass digestion issues.* Diet can rapidly reshape the microbiome within 24-48 hours, but severe dysbiosis may require microbial transplants if key microbes are extinct.* Chang’s research shows a high-fiber, low-fat diet outperforms fecal microbial transplants in restoring microbiome resilience in mice post-antibiotics.* Future microbiome medicine may involve personalized “omni microbial transplants” targeting both small and large intestines for comprehensive restoration.Related episode:* M&M 203: Metagenomics, Microbiome Transmission, Gut Microbiome in Health & Disease | Nicola Segata*Not medical advice.* Full audio version: [Apple] [Spotify] [Elsewhere]* Full video version: [YouTube] * Support M&M if you find value in this content.* Episode transcript below.Episode Chapters:00:00:00 Intro00:01:22 Guest Introduction & Microbiome Overview00:07:29 Antibiotics & Microbiome Health00:12:27 Defining Dysbiosis & Microbiome Functions00:19:39 Functional Markers & Microbiome Testing00:25:48 Case Study: Antibiotic-Induced Gut Issues00:33:20 Dietary Fiber Types & Gut Health00:41:54 Western Diets & Microbiome Damage00:49:52 Microbiome Resilience & Recovery00:56:55 Fecal Microbial Transplants & Future Therapies01:04:14 Diet-Driven Microbiome Changes01:12:10 Current Research & Future Directions01:22:51 Closing Thoughts & Gut-Brain AxisFull AI-generated transcript below. Beware of typos & mistranslations!

Sleep Duration, Sleep Timing & Short-Sleepers | Ying-Hui Fu | 239
This is a free preview of a paid episode. To hear more, visit mindandmatter.substack.comWide release date: July 13, 2025Episode Summary: Dr. Ying-Hui Fu discusses her research on the genetics of sleep, focusing on natural short sleepers who thrive on 4-6 hours of sleep and the heritability of sleep traits like duration and timing. She explores how sleep efficiency, rather than just duration, may explain why some need less sleep without health deficits, and delves into the molecular and circadian mechanisms regulating sleep. Fu also highlights the societal impact of poor sleep, the challenges of modern lifestyles, and her lab’s efforts to translate findings into interventions that could enhance sleep efficiency for better health.About the guest: Ying-Hui Fu, PhD is a neuroscientist and human geneticist who earned her PhD in biochemistry and molecular biology, focusing on human neurological and neurodegenerative diseases. She is a professor at the University of California, San Francisco, where her lab studies the genetics of human sleep behaviors, particularly sleep duration and schedule.Discussion Points:* Sleep Traits Are Genetic: Sleep duration (how long you sleep) and sleep schedule (when you sleep) are partially heritable, with most people needing 7-9 hours, while rare natural short sleepers thrive on 4-6 hours.* Natural Short Sleepers: These individuals fall asleep quickly, have high sleep efficiency, and live healthy, active lives without deficits, possibly due to faster toxin clearance and repair during sleep.* Sleep Efficiency Matters: Short sleepers may complete restorative sleep processes (e.g., clearing toxins, repairing damage) more efficiently, allowing them to need less sleep.* Circadian & Entrainment Pathways: Sleep timing is regulated by a molecular clock and environmental cues like light, with mutations in entrainment pathways causing extreme schedules (e.g., early bedtime or night owl tendencies).* Modern Lifestyle Harms Sleep: Stimuli like blue light and tense media disrupt sleep patterns, leading to widespread sleep deprivation and health risks.* Shift Work Risks: Shift workers face higher risks of diseases like cancer and neurodegeneration due to disrupted sleep cycles, though genetic background influences susceptibility.* Finding Your Sleep Rhythm: A two-week vacation without stimulants or artificial light can help determine your natural sleep schedule and duration.* Sleep’s Health Impact: Poor sleep is a major factor in aging and diseases like Alzheimer’s, making it as critical as diet and exercise for health.* Future Research Goals: Fu’s lab aims to identify converging molecular and neural pathways to enhance sleep efficiency, potentially reducing disease rates and boosting economic productivity.Related episode:* M&M 237: Circadian Biology: Genetics, Behavior, Metabolism, Light, Oxygen & Melatonin | Joseph Takahashi*Not medical advice.* Full audio version: [Apple] [Spotify] [Elsewhere]* Full video version: [YouTube] * Support M&M if you find value in this content.* Episode transcript below.Episode Chapters:00:00:00 Intro00:01:22 Guest Background & Lab Focus00:02:45 Sleep Traits & Genetics00:06:03 Natural Short Sleepers00:11:45 Sleep Efficiency & Architecture00:14:25 Genetic Mutations & Mouse Models00:20:09 Glymphatic System & Waste Clearance00:22:08 Factors Affecting Sleep Duration00:26:24 Sleep Timing & Circadian Rhythms00:32:56 Why Sleep Variation Exists00:34:52 Interventions for Short Sleep00:38:40 Modern Technology & Sleep Disruption00:42:07 Sleep, Aging & Neurodegeneration00:44:11 Demyelinating Diseases & Research Shift00:45:41 Current Projects & Short Sleeper Traits00:49:13 Importance of Sleep for HealthFull AI-generated transcript below. Beware of typos & mistranslations!

Hormones, Endocrine Disruptors & the Fate of Modern Liberal Society | Charles Cornish-Dale | 238
This is a free preview of a paid episode. To hear more, visit mindandmatter.substack.comWide release date: July 8, 2025Episode Summary: Dr. Charles Cornish-Dale discusses the decline of masculinity in modern society, linking it to falling testosterone levels, environmental endocrine disruptors, and the limitations of liberal democracy. Drawing on Francis Fukuyama’s “End of History & the Last Men” and historical perspectives, Cornish-Dale argues that biological and societal factors, including diet and hormonal interventions like birth control, are reshaping male and female behaviors, with profound implications for health and social structures.About the guest: Charles Cornish-Dale, PhD is a medieval historian and anthropologist with a PhD from Oxford. His new book is, “The Last Men: Liberalism and the Death of Masculinity.”Discussion Points:* Thymos & Masculinity: Cornish-Dale uses the ancient Greek concept of thymos, meaning spiritedness, to explain male drives for recognition and distinction, which he ties to testosterone-driven behaviors.* Testosterone Decline: Studies like the Massachusetts Male Aging Study show a ~20% drop in male testosterone levels over 17 years, correlating with reduced reproductive health and social withdrawal.* Endocrine Disruptors: Chemicals in plastics, pesticides, and soy products mimic estrogen, disrupting hormonal balance and potentially causing developmental and behavioral issues.* Diet & Behavior: Historical shifts to grain-based diets, as noted by Plato, and modern plant-based trends may suppress thymos and alter hormonal profiles, impacting societal dynamics.* Hormonal Contraceptives: Birth control can thin the ventromedial prefrontal cortex in women, affecting emotional regulation, especially if taken during teenage years, with potential permanent effects.* Fukuyama’s End of History Framework: Cornish-Dale critiques liberal democracy’s inability to satisfy megalothymia (the desire to be better), contributing to a crisis of purpose for men.* Historical Context: The agricultural revolution led to smaller, sicker populations, possibly due to coerced labor and dietary changes, highlighting diet’s role in shaping biology and behavior.Related episode:* M&M 193: History of Diet & Food, Population Density & Social Stability, Psychological Pandemics, Physical & Mental Health in Civilizational Cycles*Not medical advice.* Full audio version: [Apple] [Spotify] [Elsewhere]* Full video version: [YouTube] * Support M&M if you find value in this content.* Episode transcript below.Episode Chapters:00:00:00 Intro00:01:29 Guest Background & Book Overview00:03:26 Thymos Concept & Ancient Greeks00:07:10 Fukuyama & End of History00:10:23 Crisis of Masculinity & Last Man00:16:56 Mind-Body Dualism & Western Thought00:24:33 Testosterone Effects & Misconceptions00:30:04 Estrogen, Diet & Behavior00:44:25 Testosterone Decline & Studies00:51:57 Endocrine Disruptors & Environment01:01:30 Diet, History & Social Control01:12:55 Agricultural Revolution & Genetics01:14:38 Hormonal Contraceptives & Women01:24:09 William James & War with Nature01:29:19 Final Thoughts & Biology’s RoleFull AI-generated transcript below. Beware of typos & mistranslations!

Circadian Biology: Genetics, Behavior, Metabolism, Light, Oxygen & Melatonin | Joseph Takahashi | 237
This is a free preview of a paid episode. To hear more, visit mindandmatter.substack.comWide release date: July 4, 2025Episode Summary: Dr. Joseph Takahashi discusses circadian rhythms, exploring their biological basis, from molecular mechanisms to their impact on metabolism and health; the discovery of circadian clock genes; role of the suprachiasmatic nucleus, and how light, feeding, and oxygen influence these rhythms. The conversation highlights practical implications, such as the effects of artificial light and meal timing on health, and touches on emerging research linking stronger circadian clocks to longevity.About the guest: Joseph Takahashi, PhD is a renowned neuroscientist at UT Southwestern Medical Center, where he leads research on circadian clock genes.Discussion Points:* The suprachiasmatic nucleus in the hypothalamus acts as the brain's central clock, syncing with light via the retina.* Key circadian genes like CLOCK and BMAL regulate thousands of genes, especially those involved in metabolism, impacting health outcomes.* Internal desynchronization, when brain and organ clocks misalign (e.g., from eating at night), can lead to metabolic issues like pre-diabetes.* In mice, eating at the right time (night for nocturnal animals) extends lifespan by up to 35% under caloric restriction, compared to 10% with spread-out feeding.* Artificial light, especially blue light at night, disrupts melatonin and circadian rhythms, while natural sunlight supports healthy eye development.* Melatonin, a darkness-signaling hormone, is best for resetting rhythms (e.g., jet lag) at low doses, not as a sedative, and U.S. supplements vary widely in quality.* Oxygen-sensing proteins interact with circadian clock components, hinting at links between altitude, metabolism, and health.* Learning and memory show diurnal variations, with better performance at certain times, influenced by circadian modulation of synaptic activity.* A stronger circadian clock, created genetically in mice, led to 16% longer lifespan and resistance to weight gain (unpublished research).Related episode:* M&M 202: Why Do Animals Sleep? | Vlad Vyazovskiy*Not medical advice.* Full audio version: [Apple] [Spotify] [Elsewhere]* Full video version: [YouTube] * Support M&M if you find value in this content.* Episode transcript below.Episode Chapters:00:00:00 Intro00:03:01 Circadian Rhythms & Ecology00:09:29 Suprachiasmatic Nucleus & Central Clock00:15:18 Clock Gene Discovery00:23:23 Molecular Clock Dynamics00:32:44 Clock Genes & Metabolism00:41:18 Circadian Mutations & Obesity00:48:13 Internal Desynchronization & Health00:56:07 Artificial Light & Photobiology01:04:06 Melanopsin & Circadian Photoreceptors01:13:20 Oxygen & Circadian Regulation01:19:36 Circadian Rhythms & Learning01:24:49 Morning vs. Evening People01:29:20 Melatonin & Its Functions01:39:37 Practical Circadian Health Tips01:47:07 Boosting Clock Strength & LongevityFull AI-generated transcript below. Beware of typos & mistranslations!

Cell Biology: Cytoskeleton, Self-Assembly & Self-Organization, Wound Healing | Bill Bement | 236
This is a free preview of a paid episode. To hear more, visit mindandmatter.substack.comWide release date: June 29, 2025Episode Summary: Cell Biologist Dr. Bill Bement explains the dynamic world of the cell cortex, discussing how actin filaments and microtubules drive processes like cell division and wound healing through self-assembly and self-organization; energy dynamics of these processes; the role of rho GTPases in patterning; the implications for diseases such as cancer and muscular dystrophy, using vivid analogies and video demonstrations to make complex concepts accessible.About the guest: Bill Bement, PhD is a cell biology professor at the University of Wisconsin-Madison, where he has studied cellular processes for over 30 years. He leads a lab focused on the cell cortex, investigating cell division and repair. His work emphasizes self-organization and cytoskeletal dynamics, contributing to insights into diseases like muscular dystrophy.Discussion Points:* The cell cortex, the outer layer of a cell, includes the plasma membrane and underlying proteins like actin and myosin, which enable dynamic shape changes.* Actin filaments self-assemble without energy input, growing and shrinking to facilitate cell movement and division, while microtubules, stiffer hollow tubes, aid in chromosome separation.* Self-organization in cells, driven by energy-dependent feedback loops, creates complex patterns like mitotic spindles, as shown by experiments with DNA-coated beads forming spindle-like structures.* Cellular wound healing involves concentric rings of rho GTPases and actin, closing wounds rapidly, a process critical for surviving natural damage from mechanical stress or toxins.* Energy costs of cytoskeletal rearrangements are significant but likely less than protein synthesis, though precise measurements remain challenging.* Cancer metastasis may rely on enhanced cell repair, allowing metastatic cells to survive mechanical damage while squeezing through tissues.* Muscular dystrophy involves excessive damage or impaired repair, highlighting the importance of cell repair mechanisms.* Bement’s lab is developing tools for synthetic self-organization, aiming to manipulate cellular processes to address repair deficits in diseases.Related episode:* M&M 220: Cell Death, Oxidative Stress, PUFAs & Antioxidants | Pamela Maher*Not medical advice.* Full audio version: [Apple] [Spotify] [Elsewhere]* Full video version: [YouTube] * Support M&M if you find value in this content.* Episode transcript below.Episode Chapters:00:00:00 Intro00:01:22 Guest Introduction & Background00:04:05 Cell Cortex & Cytoskeleton Overview00:09:26 Actin & Myosin Dynamics00:15:31 Microtubules & Structural Roles00:20:09 Self-Assembly Mechanisms00:27:29 Self-Organization & Emergent Patterns00:34:09 Cellular Patterning & Rho GTPases00:41:33 Wound Healing Visualization00:49:43 Cell Division & Rho Waves00:57:03 Protein Localization & Pattern Formation01:04:54 Energy Costs of Cytoskeletal Processes01:10:58 Membrane Damage & Lipid Peroxidation01:17:39 Cancer & Enhanced Cell Repair01:22:00 Current Lab Research & Synthetic Self-Organization01:27:08 Final ThoughtsFull AI-generated transcript below. Beware of typos & mistranslations!

Computational Neuroscience, Machine Learning vs. Biological Learning, Large Language Models | Marius Pachitariu | 235
This is a free preview of a paid episode. To hear more, visit mindandmatter.substack.comWide release date: June 25, 2025Episode Summary: Dr. Marius Pachitariu discusses how the brain computes information across scales, from single neurons to complex networks, using mice to study visual learning. He explains the differences between supervised and unsupervised learning, the brain’s high-dimensional processing, and how it compares to artificial neural networks like large language models. The conversation also covers experimental techniques, such as Neuropixels probes and calcium imaging, and the role of reward prediction errors in learning.About the guest: Marius Pachitariu, PhD is a group leader at the Janelia Research Campus, leading a lab focused on neuroscience with a blend of experimental and computational approaches.Discussion Points:* The brain operates at multiple scales, with single neurons acting as computational units and networks creating complex, high-dimensional computations.* Pachitariu’s lab uses advanced tools like calcium imaging to record from tens of thousands of neurons simultaneously in mice.* Unsupervised learning allows mice to form visual memories of environments without rewards, speeding up task learning later.* Brain activity during sleep or anesthesia is highly correlated, unlike the high-dimensional, less predictable patterns during wakefulness.* The brain expands sensory input dimensionality (e.g., from retina to visual cortex) to simplify complex computations, a principle also seen in artificial neural networks.* Reward prediction errors, driven by dopamine, signal when expectations are violated, aiding learning by updating internal models.* Large language models rely on self-supervised learning, predicting next words, but lack the forward-modeling reasoning humans excel at.Related episode:* M&M 44: Consciousness, Perception, Hallucinations, Selfhood, Neuroscience, Psychedelics & "Being You" | Anil Seth*Not medical advice.* Full audio version: [Apple] [Spotify] [Elsewhere]* Full video version: [YouTube] * Support M&M if you find value in this content.* Episode transcript below.Episode Chapters:00:00:00 Intro00:05:25 Neural Computations & Scales00:13:30 Single Neuron Computations00:21:35 Network Dynamics & Complexity00:30:33 Recording Techniques & Tools00:39:32 Brain Efficiency & Metabolism00:47:30 Population Activity & Correlations00:56:10 High-Dimensional Brain Activity01:03:46 Supervised & Unsupervised Learning01:12:37 Experimental Paradigm & Mouse Behavior01:22:29 Visual Memory & Neural Changes01:34:58 LLMs & Brain Reasoning Comparison01:45:01 Closing Thoughts & Future DirectionsFull AI-generated transcript below. Beware of typos & mistranslations!

Brain Fat: Dietary PUFAs, Cannabinoids, Fatty Acid Binding Proteins, Anxiety & Mood | Steven Laviolette | 234
This is a free preview of a paid episode. To hear more, visit mindandmatter.substack.comWide release date: June 16, 2025Episode Summary: Dr. Steven Laviolette discusses the role of lipids, particularly endocannabinoids and fatty acids, in brain signaling and mental health, discussing how dietary omega-3 and omega-6 imbalances, prenatal THC exposure, and fatty acid binding proteins (FABPs) impact mood, anxiety, and neuroinflammation, while highlighting potential dietary interventions to mitigate these effects.About the guest: Steven Laviolette, PhD is a professor at the University of Western Ontario, holding a PhD, and his lab focuses on the brain’s cannabinoid system and its role in mental health. He investigates how cannabinoids and fatty acids influence brain development and disorders like anxiety and schizophrenia.Discussion Points:* The brain uses fats like endocannabinoids (e.g., anandamide, 2-AG) not just for structure but as signaling molecules to regulate emotional and cognitive processing.* Prenatal THC exposure in rodents skews the omega-3/omega-6 balance toward pro-inflammatory omega-6, leading to cognitive and mood issues in offspring, but omega-3 supplementation can mitigate these effects.* Fatty acid binding proteins (FABPs), especially FABP5, shuttle endocannabinoids like anandamide to degradation sites, and inhibiting FABP5 increases anandamide levels, reducing anxiety and depression without addictive side effects.* FABP5 inhibitors show promise as safer alternatives to benzodiazepines.* Both THC and CBD can induce problems during early development, challenging the notion that CBD is universally safe, especially during pregnancy.* Dietary interventions, like omega-3 supplementation or antioxidants (e.g., L-theanine, N-acetylcysteine), may counteract the negative psychiatric effects of cannabinoid exposure, particularly during adolescence.* The North American diet, high in omega-6, contributes to neuroinflammation and mental health issues, underscoring the need for nutritional neuroscience to address these imbalances.Related episode:* M&M 165: PUFAs in Brain Health & Disease, Dietary Fats, Brain Lipids, Nutrition | Richard Bazinet*Not medical advice.* Full audio version: [Apple] [Spotify] [Elsewhere]* Full video version: [YouTube] [Rumble]* Support M&M if you find value in this content.* Episode transcript below.Episode Chapters:00:00:00 Intro00:02:08 Lipids & Brain Signaling00:10:12 Neuroinflammation & Mood Disorders00:14:18 Prenatal THC & Omega-3/6 Balance00:18:01 Sex Differences in THC Effects00:22:48 Fatty Acid Binding Proteins00:28:16 FABP5 Inhibitors & Anxiety00:33:52 FABP5 Functions & Knockout Effects00:38:33 Dietary Interventions & Brain Development00:43:21 FABP5 & Other Fatty Acids00:48:17 Nutrition & Psychiatric Disorders00:52:43 THC, CBD, & Inflammation00:59:15 THC vs. CBD Effects01:04:44 Future Research & Dietary InterventionsFull AI-generated transcript below. Beware of typos & mistranslations!

Feel the Burn: Seed Oils, Memes & Oxidative Stress | Brian Kerley | 233
This is a free preview of a paid episode. To hear more, visit mindandmatter.substack.comWide release date: June 11, 2025Episode Summary: Dr. Brian Kerley talks about the health impacts of seed oils, their high omega-6 fatty acid content, and their role in chronic diseases through mechanisms like lipid peroxidation and oxidative stress. They explore how these industrially processed oils may contribute to conditions like heart disease and obesity, discuss the challenges of studying their long-term effects, and highlight the cultural and political dimensions of dietary trends.About the guest: Brian Kerley, MD is a family medicine-trained hospitalist physician who gained online prominence as the "Seed Oil Disrespecter" through his meme account, raising awareness about the health risks of seed oils.Discussion Points:* Seed Oils & Health Risks: Seed oils (e.g., soybean, corn, etc.) are high in omega-6 polyunsaturated fatty acids (PUFAs), linked to oxidative stress and lipid peroxidation, producing toxic compounds like 4-HNE and acrolein that damage cells.* Evolutionary Perspective: The high omega-6 levels in modern diets are evolutionarily novel, deviating from natural omega-6 to omega-3 ratios found in traditional diets, potentially exacerbating health issues across diverse populations.* Challenges with RCTs: Randomized controlled trials (RCTs) often fail to capture the long-term effects of seed oils due to the need for extended washout periods (up to 8 years) and the pervasive presence of omega-6 in modern food environments.* Cultural & Political Coding: Dietary trends like seed oil avoidance have become politically charged, with Kerley noting the polarization between institutional health narratives and alternative health communities, complicating public health discussions.* Personal Impact: Dr. Kerley’s focus on seed oils stems from personal experiences, including managing his daughter’s mitochondrial disorder, highlighting the personal stakes in dietary choices.Related episode:* M&M 192: Seed Oils, Chronic Disease, Diet & Religious Cults, Mainstream Medicine vs. Independent Research | Tucker Goodrich*Not medical advice.* Full audio version: [Apple] [Spotify] [Elsewhere]* Full video version: [YouTube] [Rumble]* Support M&M if you find value in this content.* Episode transcript below.Episode Chapters:00:00:00 Intro00:01:31 Guest Background & Role00:04:58 Nutrition Interest Origins00:10:25 Seed Oils & Taste Impact00:13:03 Defining Seed Oils00:16:36 Evolutionary Perspective on Omega-6 PUFAs00:23:29 Seed Oil Focus Journey00:31:56 Lipid Peroxidation & Health Risks00:40:12 Cellular Damage Mechanisms00:46:37 RCT Limitations & ToxinsFull AI-generated transcript below. Beware of typos & mistranslations!

SSRIs, Sexual Dysfunction, Suicide & Mass Shootings, How FDA Works | David Healy | 232
This is a free preview of a paid episode. To hear more, visit mindandmatter.substack.comWide release date: June 5, 2025Episode Summary: Dr. David Healy critiques modern medicine, focusing on SSRIs and psychiatric medicine, including: how pharmaceutical companies manipulate clinical trial data, ghostwrite studies, and influence medical practice, often ignoring patient experiences; highlighting issues like post-SSRI sexual dysfunction (PSSD), the immediate sensory effects of SSRIs, and their potential to induce suicidal or violent behavior; challenging the reliance on randomized controlled trials (RCTs) over individual patient reports; and more.About the guest: David Healy, MD, PhD, a psychiatrist and psychopharmacologist, has decades of experience researching the serotonin system and SSRIs, working across Ireland, the UK, Canada, and the US. He is a professor at McMaster University and a vocal critic of pharmaceutical industry practices.Discussion Points:* SSRIs cause near-immediate sensory effects, like genital numbing, in most people.* Post-SSRI sexual dysfunction (PSSD) can persist for years or decades after stopping the drug, affecting many long-term users.* Healy argues RCTs prioritize averages over individual experiences, often missing serious side effects like suicidality.* Pharmaceutical companies ghostwrite studies and manipulate data, with journals like the New England Journal of Medicine publishing misleading articles.* Serotonin theory of depression lacks evidence.* Industry tactics include dismissing patient reports as anecdotes and using high doses in trials to mask weak efficacy.* SSRIs can increase suicide risk, not just during initiation but also when adjusting doses or withdrawing, as seen in cases like the Aurora movie theater shooting.* Regulatory bodies like the FDA often fail to investigate adverse effects due to bureaucratic processes and lack of follow-up.* Healy emphasizes doctors’ failure to prioritize patient observations, driven by industry-influenced standards of care.Related episode:* M&M 88: Depression, Serotonin, SSRIs, Psychiatry & Social Media | Joanna Moncrieff*Not medical advice.* Full audio version: [Apple] [Spotify] [Elsewhere]* Full video version: [YouTube] [Rumble]* Support M&M if you find value in this content.* Episode transcript below.Episode Chapters:00:00:00 Intro00:04:41 Critique of Western History Narrative00:09:19 Internet & Information Democratization00:15:14 SSRI Development & Medical Observation00:23:42 Early SSRI Research & Patient Insights00:31:11 Nature of Evidence in Medicine00:37:52 Immediate SSRI Effects & Misconceptions00:45:16 Acute SSRI Sensory Impacts00:53:13 Long-term SSRI Side Effects01:00:31 SSRI Trial Design & Industry Tactics01:09:32 Data Manipulation in Drug Approval01:16:15 Post-SSRI Sexual Dysfunction (PSSD)01:24:47 PSSD Manifestations & Prevalence01:36:19 SSRI Withdrawal Challenges01:44:02 Ghostwriting & Industry Influence01:51:41 SSRIs, Suicide, & Violence02:03:01 Final Thoughts on Psychiatric MedicineFull AI-generated transcript below. Beware of typos & mistranslations!

Psychedelic Chemistry, Magic Mushrooms, Kratom, Gray Market Research Chemicals, Vape Shops | Andrew Chadeayne | 231
This is a free preview of a paid episode. To hear more, visit mindandmatter.substack.comWide release date: May 25, 2025Episode Summary: CaaMTech CEO Dr. Andrew Chadeayne, discusses the chemistry of magic mushrooms and other psychedelics, their potential therapeutic applications, and the challenges of commercializing them in a shifting biotech market. The conversation delves into the complexities of magic mushroom composition, debunking myths like "lemon tekking," and uncovers the booming, unregulated alternative products industry, including kratom and synthetic psilocybin analogs sold in vape shops.About the guest: Andrew Chadeayne, PhD is a chemist who also holds a law degree, specializing in patent law. He is the founder and CEO of CaaMTech, a startup specializing in the development of novel psychedelic compounds. Chadeayne collaborates with academic labs to advance psychedelic science while navigating the commercial landscape.Discussion Points:* Psilocybin is a prodrug converted to psilocin, but magic mushrooms contain other psychoactive molecules like beta-carbolines, which may enhance effects by inhibiting degradation.* Chadeayne’s team develops psilocin prodrugs to shorten psychedelic trips, making clinical administration more manageable without altering effects.* The "lemon tek" myth—that lemon juice converts psilocybin to psilocin—is false; it enhances absorption by extracting compounds, not altering chemistry.* Magic mushroom bruising indicates psilocin degradation, as oxidized psilocin forms blue, inactive dimers.* The multi-billion dollar alternative products industry sells kratom and synthetic psilocybin analogs, often mislabeled, in vape shops, raising safety and regulatory concerns.* Kratom’s minor alkaloid, 7-hydroxymitragynine, is ~40x more potent than morphine.* Non-hallucinogenic serotonin 2A agonists, like those developed by CaaMTech’s spinout, show promise for conditions like migraines without requiring a psychedelic trip.Related episode:* M&M 71: Chemistry of Magic Mushrooms, DMT Analogues, Entourage Effects in Cannabis & Fungi, Novel Psychedelic Medicines, Psychedelic Startups | Andrew Chadeayne*Not medical advice.* Full audio version: [Apple] [Spotify] [Elsewhere]* Full video version: [YouTube] [Rumble]* Support M&M if you find value in this content.* Episode transcript below.Episode Chapters:00:00:00 Intro00:02:21 Guest Background & CaaMTech Overview00:08:55 Psychedelic Chemistry & Magic Mushroom Molecules00:15:38 Developing New Psychedelic Prodrugs00:22:14 Academic Collaborations & Science Funding00:29:13 Non-Hallucinogenic Psychedelic Derivatives00:35:53 Debunking Psilocybin Myths00:44:16 Gray Market & Magic Mushroom Chocolate Bars00:54:33 Kratom & Alternative Products Industry01:03:00 7-Hydroxymitragynine Risks & Regulation01:12:51 Industry Self-Regulation & Future Directions01:22:17 Therapeutic Potential Without the Trip01:30:29 CaaMTech’s Future & Scientific IntegrityFull AI-generated transcript below. Beware of typos & mistranslations!

Linoleic Acid, Seed Oils, mTOR & Breast Cancer | Nikos Koundouros & John Blenis | 229
This is a free preview of a paid episode. To hear more, visit mindandmatter.substack.comWide release date: May 15, 2025Episode Summary: New research on how dietary fats, particularly omega-6 fatty acids like linoleic acid, influence triple-negative breast cancer progression by activating the mTOR pathway, a key regulator of cell growth. They explore the role of the FABP5 protein in enhancing cancer cells’ sensitivity to omega-6 fats, the differences between breast cancer subtypes, and the broader implications of dietary balance for health, emphasizing the need for personalized nutrition strategies.About the guest: John Blenis, PhD is a Professor of Pharmacology and Associate Director of Basic Science at the Meyer Cancer Center at Weill Cornell Medicine. Nikos Koundouros, PhD is a postdoctoral fellow in Blenis’ lab, focusing on nutrient sensing in breast cancer.Note: Podcast episodes are fully available to paid subscribers on the M&M Substack and everyone on YouTube. Partial versions are available elsewhere. Transcript and other information on Substack.Key Conversation Points:* mTOR’s Role: mTOR pathway acts as a cellular “brain,” sensing nutrients like amino acids, glucose, and fats to regulate growth, but its dysregulation can drive cancer.* Omega-6 PUFAs & Cancer: High dietary omega-6 fatty acids, like linoleic acid found in seed oils, can fuel triple-negative breast cancer growth by activating mTOR, especially in cells with high FABP5 expression.* FABP5 as a Biomarker: FABP5, a lipid chaperone protein, is overexpressed in triple-negative breast cancer, making these tumors more sensitive to omega-6 fats, suggesting its potential as a therapeutic target.* Dietary Balance Matters: Modern diets with high omega-6 to omega-3 ratios disrupt inflammation balance, unlike historical 1:1 ratios, potentially increasing cancer risk.* Personalized Nutrition: Genetic variations and cancer subtypes highlight the need for tailored dietary recommendations, as blanket nutrition advice may not suit all patients.* Research Gaps: While omega-6 fats exacerbate existing triple-negative breast cancer, their role in initiating cancer remains unclear, requiring further study.* Broader Implications: High omega-6 intake may influence other cancers (e.g., prostate, colon) and chronic diseases like obesity, linked to FABP5 and inflammation.Related episode:* M&M 200: Dietary Fats & Seed Oils in Inflammation, Colon Cancer & Chronic Disease | Tim Yeatman & Ganesh Halade*Not medical advice.* Full audio version: [Apple] [Spotify] [Elsewhere]* Full video version: [YouTube] [Rumble]* Support M&M if you find value in this content.* Episode transcript below.Episode Chapters:00:00:00 Intro00:03:15 mTOR Overview00:11:40 Nutrient Sensing by mTOR00:19:26 Breast Cancer Subtypes00:27:07 Diet & Cancer Links00:34:56 Essential Fatty Acids00:43:16 Linoleic Acid Biochemistry00:50:35 Study on Omega-6 & Breast Cancer00:59:23 FABP5 & Omega-6 Sensitivity01:08:30 Linoleic Acid & mTOR Interaction01:15:50 Cause and Effect in Animal Models01:23:48 Omega-3 Effects & Broader Implications01:33:26 Clinical Studies & Nutrition Debates01:41:59 Final Thoughts & Future ResearchFull AI-generated transcript below. Beware of typos & mistranslations!

Rhythms, Memory, Time, Place, Representation & the Brain | György Buzsáki | 228
This is a free preview of a paid episode. To hear more, visit mindandmatter.substack.comWide release date: May 10, 2025Episode Summary: Dr. Gyorgy Buzsaki discusses the hippocampus’s role beyond memory and spatial navigation, delving into its broader functions in cognition, action planning, and brain-body interactions; how hippocampal rhythms, like sharp wave ripples, influence memory consolidation, glucose regulation, and metabolic health, challenging conventional neuroscience assumptions; the interplay of brain rhythms, sleep, and preconfigured neural dynamics; the history and conceptual foundations of neuroscience; and more.About the guest: Gyorgy Buzsaki, MD, PhD is a professor at NYU. He leads a lab investigating how neural circuits underpin cognition, particularly through oscillations and brain-body interactions. His work has significantly advanced understanding of memory formation and spatial navigation.Note: Podcast episodes are fully available to paid subscribers on the M&M Substack and everyone on YouTube. Partial versions are available elsewhere. Transcript and other information on Substack.Key Conversation Points:* Hippocampus isn’t just for memory or navigation; it may orchestrate action planning and abstract representations of the world, shaped by evolutionary constraints.* Brain rhythms, like sharp wave ripples, synchronize neural activity, enabling efficient communication and impacting bodily functions like glucose homeostasis.* Sharp wave ripples, prominent during non-REM sleep and consummatory states, are critical for memory consolidation and may link sleep disruptions to metabolic disorders.* Buzsaki challenges the idea of memory as fixed synaptic patterns, proposing it’s more like dynamic, cloud-like sequences, endlessly reconfigurable.* The brain’s intrinsic dynamics prioritize action generation and learning from consequences over external representations.Related episode:* M&M 16: Sleep, Dreams, Memory & the Brain | Bob Stickgold*Not medical advice.* Full audio version: [Apple] [Spotify] [Elsewhere]* Full video version: [YouTube] [Rumble]* Support M&M if you find value in this content.* Episode transcript below.Episode Chapters:00:03:21 Gyorgy Buzsaki Intro00:05:04 Hippocampus Functions00:10:32 Action Planning Hypothesis00:15:39 Preconfigured Constraints00:21:26 Historical Neuroscience Perspectives00:29:57 Inherited Conceptual Assumptions00:38:27 Place Cells Discovery00:49:47 Neural Energy Costs00:57:24 Brain Dynamics Maintenance01:05:04 Brain Rhythms Overview01:15:36 Gamma Oscillations Role01:22:09 Memory and Engrams01:30:45 Sharp Wave Ripples and Metabolism01:39:30 Ripple Emergence ContextsFull AI-generated transcript below. Beware of typos & mistranslations!

Biophysics of Life: Biophotons, Light, Quantum Biology, Regeneration & Cancer | Nirosha Murugan | 227
This is a free preview of a paid episode. To hear more, visit mindandmatter.substack.comEpisode Summary: Dr. Nirosha Murugan discusses the role of biophysics in biology, focusing on how light, particularly biophotons emitted by cells, influences processes like wound healing, neural activity, and cancer detection; how microtubules may act as biological fiber optics, the impact of modern light environments on health; her work inducing limb regeneration in frogs using silk hydrogels and growth factors; cancer as an energetic dysfunction; the potential of non-invasive photonic diagnostics; the need for new tools to study these phenomena.About the guest: Nirosha Murugan, PhD is a biophysicist and assistant professor at Wilfrid Laurier University in Waterloo, Ontario, focusing on how optical, magnetic, and electric signals influence biological processes. Her lab investigates bioelectricity, biophotonics, and regeneration, aiming to leverage these signals for disease detection and tissue engineering.Note: Podcast episodes are fully available to paid subscribers on the M&M Substack and everyone on YouTube. Partial versions are available elsewhere. Transcript and other information on Substack.Key Conversation Points:* Cells emit biophotons, ultra-weak light tied to metabolism, which may carry information for processes like immune response and neural communication.* Microtubules might function as biological fiber optics, potentially guiding light within cells for signaling purposes.* Red and near-infrared light can accelerate wound healing and reduce inflammation, likely by modulating mitochondrial activity.* Cancer cells emit distinct photonic signatures, which could enable non-invasive diagnostics by detecting light differences from healthy tissues.* Modern light environments, unlike natural sunlight, may subtly affect health by altering biological responses to electromagnetic signals.* Regeneration in adult frogs was induced using silk hydrogels and growth factors, hinting at ways to reawaken human regenerative potential.* Biological systems act as metamaterials, patterning energy flow in ways that constrain and shape molecular and behavioral outcomes.Related episode:* M&M 221: Regenerative Energy & the Light Inside You | Jack Kruse*Not medical advice.* Full audio version: [Apple] [Spotify] [Elsewhere]* Full video version: [YouTube] [Rumble]* Support M&M if you find value in this content.* Episode transcript below.Episode Chapters:00:00:00 Intro00:01:37 Nirosha Murugan Intro00:04:11 Light Beyond Vision00:09:07 Biophotons Explained00:15:15 Water's Biophysical Role00:20:21 Microtubules as Fiber Optics00:28:13 Microtubule Functionality00:34:35 Biophoton Detection Tools00:41:22 Optogenetics Insights00:48:17 Brain Photon Detection00:54:48 Physics of Life01:01:12 Energy in Biology01:07:45 Electromagnetic Concerns01:13:45 Regeneration Research01:19:49 Wound Healing & Light01:25:30 Cancer Photonics01:32:27 Future of BiophysicsFull AI-generated transcript below. Beware of typos & mistranslations!

Psychedelics & Cerebral Cortex: Neuroplasticity, Psilocybin, Ketamine | Alex Kwan | 226
This is a free preview of a paid episode. To hear more, visit mindandmatter.substack.comEpisode Summary: Dr. Alex Kwan discusses how psychedelics like ketamine and psilocybin induce rapid neuroplastic changes in the brain, particularly in the prefrontal cortex, contrasting their effects with traditional antidepressants like SSRIs, and exploring their potential for treating depression and chronic pain through structural and functional brain alterations.About the guest: Alex Kwan, PhD, is an associate professor of biomedical engineering at Cornell University. His lab employs advanced imaging to study how psychedelics and other drugs affect the mammalian brain.Note: Podcast episodes are fully available to paid subscribers on the M&M Substack and everyone on YouTube. Partial versions are available elsewhere. Transcript and other information on Substack.Key Discussion Points:* Ketamine & psilocybin rapidly increase dendritic spine density in the prefrontal cortex, enhancing neural connections within days, unlike SSRIs, which take weeks.* These drugs show sustained neuroplastic changes in mice, lasting weeks to months after a single dose, suggesting long-term brain rewiring.* Serotonin 2A receptor is critical for psilocybin’s neuroplastic effects, as precise genetic knockouts in adult mice eliminate spine growth.* Unlike ketamine, psilocybin activates the insula, a brain region linked to chronic pain processing, hinting at new therapeutic potential.* Both drugs induce similar gene expression patterns in areas like the prefrontal cortex and amygdala, but differ in specific regions like the insula.Reference paper: * Study: Psilocybin’s lasting action requires pyramidal cell types and 5-HT2A receptorsRelated episode:* M&M #30: Psilocybin, Ketamine, Neuroplasticity & Imaging the Brain | Alex Kwan*Not medical advice.* Full audio version: [Apple] [Spotify] [Elsewhere]* Full video version: [YouTube] [Rumble]* Support M&M if you find value in this content.* Episode transcript below.Episode Chapters:00:00:00 Intro00:04:32 Cerebral Cortex Structure00:09:00 Excitatory vs. Inhibitory Neurons00:14:20 Depression & Neuroplasticity00:19:19 SSRI Effects & Limitations00:24:34 Ketamine & Psilocybin Rapid Effects00:30:00 Dendritic Spine Plasticity00:36:01 Serotonin 2A Receptor Debate00:41:32 Ketamine vs. Psilocybin Mechanisms00:46:08 Measuring Psychedelic Effects in Mice00:52:13 Head Twitch Response Limitations00:57:10 Serotonin 2A Receptor Knockout Results01:03:19 c-Fos Mapping Insights01:09:28 Future Research DirectionsFull AI-generated transcript below. Beware of typos & mistranslations!

Nutrition Epidemiology: Fake Science? | John Speakman | 225
This is a free preview of a paid episode. To hear more, visit mindandmatter.substack.comShort Summary: The flaws of nutrition epidemiology with Dr. John SpeakmanAbout the guest: John Speakman, PhD is a professor at the University of Aberdeen and runs a lab in Shenzhen, China, focusing on energy balance, obesity, and aging. Note: Podcast episodes are fully available to paid subscribers on the M&M Substack and everyone on YouTube. Partial versions are available elsewhere. Transcript and other information on Substack.Episode Summary: Dr. John Speakman explores the pitfalls of nutrition epidemiology, a field that links diet to health outcomes like cancer and obesity but often produces contradictory results. They discuss flawed methods like 24-hour recalls and food frequency questionnaires, which rely on memory and are prone to bias, and introduce Speakman’s new tool using doubly labeled water to screen implausible dietary data. The conversation highlights systematic biases, such as under-reporting by heavier individuals, and emerging technologies like photo diaries and AI for better dietary tracking.Key Takeaways:* Nutrition epidemiology studies often contradict each other due to unreliable methods.* Common techniques like 24-hour recalls & food frequency questionnaires suffer from memory issues, portion size issues, and systematic biases, often underestimating food intake.* Heavier individuals (higher BMI) under-report food intake more, skewing associations between diet & obesity.* Speakman’s tool, based on 6,500 doubly labeled water measurements, predicts energy expenditure to flag implausible dietary survey data.* Emerging technologies, like smartphone photo diaries and AI food identification, promise more accurate dietary tracking than traditional surveys.* Randomized controlled trials, not surveys, provide the most reliable dietary insights; single-day intake surveys linked to outcomes years later are dubious.* Speakman advises ignoring most nutrition epidemiology headlines due to their inconsistency and lack of prognostic value for behavior change.Related episode:* M&M #132: Obesity Epidemic, Diet, Metabolism, Saturated Fat vs. PUFAs, Energy Expenditure, Weight Gain & Feeding Behavior | John Speakman*Not medical advice.* Full audio version: [Apple] [Spotify] [Elsewhere]* Full video version: [YouTube] [Rumble]* Support M&M if you find value in this content.* Episode transcript below.Episode Chapters:00:00:00 Intro00:03:08 Nutrition Epidemiology Overview00:08:12 Flaws in Dietary Recall Methods00:14:42 Food Frequency Questionnaire Issues00:20:12 Validation Problems in Studies00:26:22 Basal Metabolic Rate Validation00:32:10 Doubly Labeled Water Technique00:38:20 New Predictive Tool for Surveys00:43:41 Systematic Biases in Data00:50:06 Improving Data with Red Flags00:56:15 Emerging Technologies for Tracking01:02:01 Advice on Nutrition HeadlinesFull AI-generated transcript below. Beware of typos & mistranslations!

Cancer Biology: Metabolism, Mitochondria & Energy | Thomas Seyfried | 224
This is a free preview of a paid episode. To hear more, visit mindandmatter.substack.comShort Summary: Cancer’s metabolic roots with Dr. Thomas Seyfried.About the guest: Thomas Seyfried, PhD is a professor of biology at Boston College. He has researched cancer metabolism, epilepsy, and lipid biochemistry for over 40 years.Note: Podcast episodes are fully available to paid subscribers on the M&M Substack and everyone on YouTube. Partial versions are available elsewhere. Transcript and other information on Substack.Episode Summary: Dr. Thomas Seyfried discusses the mitochondrial metabolic theory of cancer, challenging the dominant somatic mutation theory. He explores how cancer cells rely on fermentation due to defective oxidative phosphorylation, drawing on Otto Warburg’s work. Seyfried explains how ketogenic diets and nutritional ketosis can starve cancer cells by limiting glucose and glutamine, while sharing evidence from nuclear transfer experiments and clinical studies. The conversation also covers environmental factors driving cancer and the importance of metabolic flexibility for prevention.Key Takeaways:* Cancer is characterized by dysregulated cell growth, but Seyfried argues it stems from mitochondrial dysfunction, not just genetic mutations.* Cancer cells ferment glucose & glutamine, unable to use fatty acids or ketones, making ketogenic diets a potential therapeutic tool.* Nuclear transfer experiments show cancer traits reside in the cytoplasm, not the nucleus, challenging the somatic mutation theory.* Environmental factors like processed foods, stress, and poor sleep disrupt mitochondrial function, increasing cancer risk.* Seyfried’s glucose-ketone index helps monitor metabolic states to manage cancer & chronic diseases.* Cancer rates are rising in younger people, possibly due to obesity, inflammation, and environmental toxins.* Metabolic flexibility, cycling between ketosis and carb-based states, may mimic ancestral patterns and reduce chronic disease risk.Related episode:* M&M #215: Cancer Metabolism: Sugar, Fructose, Lipids & Fasting | Gary Patti*Not medical advice.* Full audio version: [Apple] [Spotify] [Elsewhere]* Full video version: [YouTube] [Rumble]* Support M&M if you find value in this content.* Episode transcript below.Episode Chapters:00:00:00 Intro00:02:57 Cancer Basics00:07:41 Genetic Theory of Cancer00:10:38 Ketogenic Diet Insights00:13:42 Warburg’s Hypothesis00:20:19 Biochemical Debates00:24:08 Molecular Biology Era00:30:07 Genetic Theory Flaws00:36:41 Nuclear Transfer Evidence00:41:59 Metabolic Theory Overview00:48:31 Fermentation in Cancer00:56:00 Mitochondrial Coupling Explained01:02:05 Mitochondrial Haplotypes01:09:56 Ketogenic Diet for Cancer01:15:22 Ketones vs. Glucose Efficiency01:21:10 Metabolic Flexibility01:24:55 Causes of Mitochondrial Damage01:29:27 Light and Mitochondrial Health01:33:13 Ketogenic Diet Composition01:39:40 Cancer Trends01:44:32 Cancer Prevention Tips01:46:17 Closing RemarksFull AI-generated transcript below. Beware of typos & mistranslations!

Ketogenic Diet: Cholesterol, Plaque & Heart Heart | Matthew Budoff | 223
This is a free preview of a paid episode. To hear more, visit mindandmatter.substack.comShort Summary: Heart health and the ketogenic diet, with expert insights from a cardiologist and researcher.About the guest: Matthew Budoff, MD, is a preventive cardiologist and professor of medicine at UCLA School of Medicine.Note: Podcast episodes are fully available to paid subscribers on the M&M Substack and everyone on YouTube. Partial versions are available elsewhere. Full transcript and other information on Substack.Episode Summary: Dr. Matthew Budoff discusses cholesterol, heart disease, and his study on the ketogenic diet’s impact on lean, metabolically healthy individuals with high LDL cholesterol. He explains LDL, HDL, and triglycerides, debunking myths about their direct link to heart disease, and emphasizes the importance of coronary calcium scans to assess plaque buildup. The study found no significant plaque progression in these individuals despite elevated LDL, challenging conventional cardiology views. Budoff also covers statins, dietary cholesterol, and personalized heart health strategies.Key Takeaways:* LDL cholesterol is not a definitive predictor of heart disease; plaque buildup, assessed via coronary calcium scans, is a better indicator.* Lean metabolically healthy people on a ketogenic diet may have high LDL without increased plaque progression after one year.* Coronary calcium scans, costing ~$100, are recommended for men around age 40 and women around 45-50 to evaluate heart disease risk.* Statins effectively lower LDL and can reverse soft plaque, but may be overprescribed for those without plaque buildup.* Dietary cholesterol has a minimal impact on blood cholesterol, as the liver produces ~85% of it.* The ketogenic diet can aid weight loss and diabetes control but may cause high LDL in some lean individuals, known as lean mass hyper-responders.* Plaque progression depends more on existing plaque than LDL levels in metabolically healthy ketogenic diet followers.* Heart health varies widely due to genetics and other unknown factors, underscoring the need for personalized assessments.Related episode:* M&M #158: Ketosis & Ketogenic Diet: Brain & Mental Health, Metabolism, Diet & Exercise, Cancer, Diabetes | Dominic D'Agostino*Not medical advice.* Full audio version: [Apple] [Spotify] [Elsewhere]* Full video version: [YouTube] [Rumble]* Support M&M if you find value in this content.* Episode transcript below.Episode Chapters:00:00:00 Intro00:01:29 Guest Introduction00:02:36 Cholesterol Basics00:09:15 Heart Disease Risk Factors00:13:56 Statins and LDL Management00:18:35 Ketogenic Diet Overview00:24:43 Study Setup and Methods00:31:10 Study Results00:38:19 Heart Scan Recommendations00:44:23 Lean Mass Hyper Responders00:50:08 LDL Guidelines Critique00:55:06 Final ThoughtsFull AI-generated transcript below. Beware of typos & mistranslations!

Genetics & Neurobiology of Obesity: Hypothalamus, Leptin, Gut Hormones, Weight Loss Drugs | Giles Yeo | 222
This is a free preview of a paid episode. To hear more, visit mindandmatter.substack.comShort Summary: The science of obesity and appetite regulation, blending genetics and neurobiology with practical insightsAbout the guest: Giles Yeo, PhD is a professor of molecular neuroendocrinology at the University of Cambridge. He leads a lab studying obesity and appetite regulation.Note: Podcast episodes are fully available to paid subscribers on the M&M Substack and everyone on YouTube. Partial versions are available elsewhere. Full transcript and other information on Substack.Episode Summary: Nick interviews Dr. Giles Yeo about the genetics and neurobiology of obesity, starting with the discovery of leptin in the obese mouse model, detailing its role in appetite regulation via the hypothalamus, and discussing GLP-1 drugs like Ozempic for weight loss. It delves into how genetic factors, like the leptin-melanocortin pathway, influence hunger, the heritability of body weight, and societal factors driving the obesity epidemic, emphasizing the interplay of biology and environment.Key Takeaways:* Leptin, discovered through the obese mouse, signals fat levels to the brain, but its absence causes severe obesity and infertility, as seen in rare human mutations.* The hypothalamus, a key brain region, senses hormones like leptin and GLP-1, balancing hunger and satiety through POMC (anorexigenic) and AgRP (orexigenic) neurons.* Body weight heritability is 40-70% at the population level, but this does not mean that 40-70% of someone’s body fat composition is due to genetic factors outside human control. Dr. Yeo unpacks how to think about it.* GLP-1 drugs (e.g., Ozempic) mimic gut hormones to reduce appetite, offering some people 15-25% weight loss, but require long-term safety monitoring.* According to Dr. Yeo, obesity reflects energy imbalance, but nutritional density matters more than calorie counting for health, and societal changes are needed to prevent it.Related episode:* M&M #132: Obesity Epidemic, Diet, Metabolism, Saturated Fat vs. PUFAs, Energy Expenditure, Weight Gain & Feeding Behavior | John Speakman*Not medical advice.* Full audio version: [Apple] [Spotify] [Elsewhere]* Full video version: [YouTube] [Rumble]* Support M&M if you find value in this content.* Episode transcript below.Episode Chapters:00:00:00 Intro00:05:04 Discovery of Leptin00:10:32 Hypothalamus Overview00:16:23 Leptin Source and Function00:22:26 Peptides vs. Neurotransmitters00:28:35 Nutrient Sensing in Hypothalamus00:35:43 Metabolism & Leptin Pathway00:42:46 Genetics of Obesity00:51:37 Heritability & Environment00:56:23 GLP-1 & Incretin Hormones01:03:41 Synthetic GLP-1 Drugs01:09:55 Drivers of Overeating01:17:59 Leptin Resistance Debate01:23:47 Current Research & Future Questions01:29:57 Closing ThoughtsFull AI-generated transcript below. Beware of typos & mistranslations!

Cell Death, Oxidative Stress, PUFAs & Antioxidants | Pamela Maher | 220
This is a free preview of a paid episode. To hear more, visit mindandmatter.substack.comShort Summary: A deep dive into the science of oxidative stress, antioxidants, aging, and neurodegenerative diseases like Alzheimer’s.About the guest: Pamela Maher, PhD is a professor at the Salk Institute in San Diego, California, with a focus on neurodegenerative diseases, oxidative stress, and antioxidants.Note: Podcast episodes are fully available to paid subscribers on the M&M Substack and everyone on YouTube. Partial versions are available elsewhere. Full transcript and other information on Substack.Episode Summary: Nick interviews Dr. Pamela Maher about her research on cell death mechanisms, including apoptosis and ferroptosis, and their roles in health and neurodegenerative diseases. They discuss how oxidative stress from reactive oxygen species can damage cells, the body’s antioxidant defenses like glutathione, and Maher’s studies on natural compounds—fisetin, curcumin, and cannabinol (CBN)—that may protect against brain cell loss. The conversation covers CBN’s mitochondrial benefits in aging mouse models and broader risk factors for dementia, like obesity and hearing loss, emphasizing early intervention.Key Takeaways:* Apoptosis is a controlled cell death process vital for development, while ferroptosis, linked to oxidative stress and iron, causes cells to burst and is tied to diseases like Alzheimer’s.* Oxidative stress arises when reactive oxygen species overwhelm antioxidants, damaging DNA, proteins, and cell membranes, especially polyunsaturated fats.* Glutathione, a key antioxidant, relies on cysteine and declines with age, but supplements like N-acetylcysteine might help, though tolerability varies.* Cannabinol (CBN), a THC byproduct, protects mitochondria and improves memory in aging mice, hinting at therapeutic potential beyond cannabinoid receptors.* Middle-age risk factors like visceral obesity, type 2 diabetes, and hearing loss significantly increase dementia risk, but hearing aids can reduce it.* Maher’s team is refining natural compounds and exploring how they might address both brain and systemic health to combat dementia.Related episode:* M&M #165: PUFAs in Brain Health & Disease, Dietary Fats, Brain Lipids, Nutrition | Richard Bazinet*Not medical advice.* Full audio version: [Apple] [Spotify] [Elsewhere]* Full video version: [YouTube] [Rumble]* Support M&M if you find value in this content.* Episode transcript below.Episode Chapters:00:00:00 Intro00:05:02 Cell Death Pathways00:10:21 Apoptosis Mechanisms00:15:10 Oxidative Stress Basics00:20:23 Antioxidant Systems00:25:56 Oxidative Damage Effects00:30:00 Ferroptosis Process00:35:04 Ferroptosis Triggers00:40:15 Lipid Peroxidation00:45:20 Therapeutic Approaches00:50:30 Natural Products Research00:55:45 Cannabinoid Studies01:00:57 Compound Benefits01:05:18 Cannabinol Effects01:10:28 Mitochondrial Protection01:15:07 Mouse Model Results01:20:09 Clinical Trial Potential01:25:41 Antioxidant Strategies01:30:06 Current Research Directions01:35:00 Risk Factors for DementiaFull AI-generated transcript below. Beware of typos & mistranslations!

Chemical Exposures, Neurodegenerative Disease & Exposomics | Gary Miller | 218
This is a free preview of a paid episode. To hear more, visit mindandmatter.substack.comShort Summary: How environmental chemical exposures affect neurodegenerative disease and health.About the guest: Gary Miller, PhD is a Professor of Environmental Health Sciences at Columbia University. His research bridges pharmacology, toxicology, and neuroscience to explore neurodegeneration, particularly Parkinson’s and Alzheimer’s diseases.Note: Podcast episodes are fully available to paid subscribers on the M&M Substack and everyone on YouTube. Partial versions are available elsewhere. Full transcript and other information on Substack.Episode Summary: Dr. Gary Miller talks about neurodegenerative diseases like Parkinson’s and Alzheimer’s, exploring their age-driven nature, early detection challenges, and environmental risk factors such as pesticide exposure and head trauma. They discuss the emerging field of exposomics—studying lifetime chemical exposures—and its potential to uncover links between pollutants, diet, and disease, while highlighting protective factors like caffeine and nicotine, and the role of technology in advancing prevention and treatment.Key Takeaways:* Neurodegenerative diseases like Parkinson’s & Alzheimer’s worsen with age, but early intervention could slow progression & improve quality of life.* Parkinson’s symptoms often appear after significant dopamine neuron loss, but subtle signs like handwriting changes or gait issues might be detectable earlier.* Environmental factors, including pesticides & air pollution, increase neurodegeneration risk, while caffeine and nicotine surprisingly lower Parkinson’s incidence.* The exposome—total lifetime exposure to chemicals—offers a new lens to study health, revealing how pollutants & diet interact with our biology.* Ultra-processed foods & common chemicals like PFAS (in packaging) raise exposure levels, but simple steps like air filters or less processed diets can help reduce risks.Related episode:* M&M #145: Epigenetics, Hormones, Endocrine Disruptors, Microplastics, Xenoestrogens, Obesogens & Obesity, Inheritance of Acquired Characteristics | Bruce Blumberg*Not medical advice.* Full audio version: [Apple] [Spotify] [Elsewhere]* Full video version: [YouTube] [Rumble]* Support M&M if you find value in this content.* Episode transcript below.Episode Chapters:00:00:00 Intro00:05:34 Early Detection of Neurodegeneration00:10:16 Parkinson’s: Dopamine & Beyond00:17:22 Parkinson’s Prevalence & Risk Factors00:25:34 Protective Effects of Caffeine & Nicotine00:31:09 The Exposome Explained00:38:02 Exposome Studies & Parkinson’s00:46:33 In Utero Exposures & Alzheimer’s Risks00:54:19 Diet, Ultra-Processed Foods & Exposures01:01:29 Participatory Research and Common Chemicals01:08:02 Reducing Everyday Exposures01:15:22 Pharmaceuticals & Neurodegeneration01:23:26 Variability in Detoxification01:29:12 Future of ExposomicsFull AI-generated transcript below. Beware of typos & mistranslations!

Cancer Metabolism: Sugar, Fructose, Lipids & Fasting | Gary Patti | 215
Short Summary: How dietary sugar (fructose) affects the growth rate of cancer.About the guest: Gary Patti, PhD is a professor at Washington University in St. Louis, holding appointments in chemistry, medicine, and geneticsNote: Podcast episodes are fully available to paid subscribers on the M&M Substack and everyone on YouTube. Partial versions are available elsewhere. Full transcript and other information on Substack.Episode Summary: Nick Jikomes talks to Dr. Gary Patti, exploring how cancer cells metabolize sugars like glucose and fructose, focusing on a recent study showing fructose indirectly boosts tumor growth in mice via liver-produced lipids called lysophosphatidylcholines (LPCs). The discussion covers cancer biology basics, the Warburg effect, tumor microenvironments, and the systemic metabolic impacts of cancer, while also touching on dietary implications, fasting, and the complexities of nutrient utilization in cancer progression.Key Takeaways:* Cancer cells often rely heavily on glucose, excreting it as lactate even when oxygen is available (Warburg effect), but take up more than their mitochondria can handle.* In a study, high fructose diets accelerated tumor growth in mice by 4x, not because cancer cells use fructose directly, but because the liver converts it to LPCs, which tumors use to build membranes.* Tumors are not just cancer cells; they recruit healthy cells in their microenvironment, and their metabolic effects ripple across the entire body, altering distant tissues.* Excessive fructose consumption (e.g., from soda, not fruit) may worsen tumor growth, but cutting it poses little risk and could benefit cancer patients, pending human studies.* Fasting may reduce cancer initiation risk in animals, but its effect on existing tumors is less clear and could worsen wasting (cachexia) in late stages.* The body tightly regulates blood glucose via the liver, so simply cutting dietary glucose won’t starve tumors, highlighting cancer’s metabolic adaptability.Related episode:* M&M #200: Dietary Fats & Seed Oils in Inflammation, Colon Cancer & Chronic Disease | Tim Yeatman & Ganesh Halade*Not medical advice.* Full audio version: [Apple] [Spotify] [Elsewhere]* Full video version: [YouTube] [Rumble]* Support M&M if you find value in this content.* Episode transcript below.Episode Chapters:00:00:00 Intro00:05:35 Cancer Biology Basics00:11:26 Tumor Initiation and Immune Evasion00:17:13 Studying Early Cancer with Zebrafish00:23:19 Metabolic Changes in Tumors00:29:16 Tumor Microenvironment and Healthy Cells00:34:28 Glucose vs. Fructose Metabolism00:40:17 Fructose and Cancer Cell Growth00:46:04 Fructose Processing in the Body00:52:49 High Fructose Diets in Mice00:59:22 Liver’s Role in Tumor Growth01:06:06 Identifying LPCs as Key Nutrient01:12:03 LPCs and Dietary Fat Connections01:17:19 Fructose Intake Implications01:23:40 Endogenous Fructose and Drugs01:27:57 Ketogenic Diets and Cancer01:34:09 Fasting and Cancer Risk01:39:59 Cancer Phases and Diet StrategiesFull AI-generated transcript below. Beware of typos & mistranslations!Gary Patti 1:31 yeah. Name is Gary patti. I'm a professor at Washington University in St Louis. Actually wear a few different hats. I'm in a couple different departments, chemistry, medicine and genetics, and I run a relatively large lab. We study cancer and metabolism. I also serve as director of some cores at the university, the clinical research core, as well as some metabolomics and proteomics cores. And then lastly, I'll just mention that I'm also a CSO of a company that does contract research services for for different omics, multi omics technologies.Nick Jikomes 2:12 Yeah, so, so you've got quite an interdisciplinary lab. You do a lot of stuff that's related to metabolism and metabolomics. You do a lot of stuff related to cancer biology, which will probably be our focus here. Today, we want to get to a paper that I thought was very interesting and very relevant to a lot of people, you know, out in the real world, which has to do with the relationship between sugar metabolism, particular, and and tumor growth. Before we get there, I want to cover off on some basics on cancer biology and and sugar and fructose metabolism. So, you know, I'm somewhat familiar with cancer biology. I'm a biology guy by training, but I was never a cancer biology person. And sort of the cartoon that I've always had in my head, or I had when I was, like a student in college and stuff, is that, you know, cancer is a cell. You know, cancer cells are cells that are have escaped the normal cell cycle regulation, and they're growing uncontrollably, and that that is largely often a genetic thing. So, you know, I learned about oncogenes and tumor suppressor genes, and so the idea was, cancer is typically a series of mutations in specific genes that allow the cell to break free of the cell cycle. Can you just give us a general sense of, are most cancers, or is the average cancer? Whatever that means, is it lar

Gut Microbiome Plasticity | Peter Turnbaugh | 214
Short Summary: How diet shapes the gut microbiome and impacts health, with microbiologist Dr. Peter Turnbaugh breaking down the complex science.About the guest: Peter Turnbaugh, PhD is a professor of microbiology and immunology at the University of California, San Francisco, where he leads a lab studying the gut microbiome’s role in nutrition and drug response.Note: Podcast episodes are fully available to paid subscribers on the M&M Substack and everyone on YouTube. Partial versions are available elsewhere. Full transcript and other information on Substack.Episode Summary: Nick Jikomes talks to Peter Turnbaugh about the gut microbiome’s response to dietary changes, focusing on high-fat diets, ketogenic diets, and their effects on health. They discuss the pitfalls of oversimplified diet labels in research, Turnbaugh’s studies comparing plant-based and animal-based diets in humans, and how these shifts rapidly alter gut microbes and short-chain fatty acid profiles. The conversation also covers the microbiome’s role in drug metabolism, its links to inflammation and autoimmunity (like multiple sclerosis), and the potential of ketogenic diets to modulate these processes via ketone bodies like BHB.Key Takeaways:* The term “high-fat diet” in research is often misleading, as it can include high carbs and vary widely, complicating study comparisons.* In a 2014 study, switching humans to a plant-based (high-fiber) or animal-based (ketogenic, no-fiber) diet changed their gut microbiome within one day, showing its remarkable adaptability.* Ketogenic diets reduce Bifidobacterium in the gut, which may lower inflammation-linked immune cells (Th17), potentially aiding conditions like multiple sclerosis.* Short-chain fatty acids (e.g., butyrate) don’t just come from fiber; they persist even on zero-fiber ketogenic diets, hinting at alternative microbial pathways.* Gut microbes can activate or deactivate drugs, like antibiotics or digoxin, suggesting microbiomes may explain why drugs work differently across individuals.* Ketone bodies like BHB alone can mimic some ketogenic diet effects on the microbiome and immunity, simplifying research and hinting at therapeutic potential.Related episode:* M&M #203: Metagenomics, Microbiome Transmission, Gut Microbiome in Health & Disease | Nicola Segata*Not medical advice.* Full audio version: [Apple] [Spotify] [Elsewhere]* Full video version: [YouTube] [Rumble]* Support M&M if you find value in this content.* Episode transcript below.Episode Chapters:00:00:00 Intro00:05:34 High-Fat Diet Issues00:11:00 Macronutrient Mapping00:17:29 Metabolic Adaptability00:25:59 2014 Study Design00:34:01 Microbiome Flexibility00:41:03 Fatty Acid Profiles00:48:41 Bile and Gut Changes00:55:09 Inflammation Links01:00:11 Keto Diet Exploration01:07:08 BHB Impact01:14:21 Microbiome Products01:19:35 Drug Response ResearchFull AI-generated transcript below. Beware of typos & mistranslations!Peter Turnbaugh 1:30 Sure. So I'm a professor in the department of Microbiology and Immunology at the University of California in San Francisco, and our lab is kind of broadly interested in how the gut microbiome, the trillions of microbes that are found within our GI tract, influence both nutrition and pharmacology.Nick Jikomes 1:51 Yeah. And so you study lots of interesting stuff. There's many different directions we could take this conversation. I've covered the microbiome a lot on this podcast. I've covered diet stuff a lot on this podcast. I want to start off pretty high level and talk about, get you talking about something that doesn't necessarily have to do with your work specifically, but it's, it's sort of broader questions related to diet and nutrition and how it's studied in the literature, and it obviously ties into the work you do. But I want to talk about some of the different types of diets that are out there that are commonly studied, and some confusion and shortcomings maybe even the literature has in terms of how those things are studied and how they're talked about. So the points I want to start is, I'm going to keep it vague intentionally, one of the most common diets that you'll read, if you dig into the scientific literature that has to do with the diet, is the so called high fat diet. And maybe some of the listeners, listeners will know from from listening to me before, but this is one of my biggest frustrations in the literature, this term high fat diet, what can you tell us about the high fat diet, and what that term maybe doesn't tell us, or problems that come from labeling and using diets with that level of specificity?Peter Turnbaugh 3:09 Yeah, that's a great question, and kind of near and dear to my heart. I don't know if you saw but we, we had a paper a few years ago where we did a meta analysis, where we were interested in, kind of comparing the microbiome profiles, mostly in mice, but also a little bit in humans. You know, across the hundreds of studies where people have fed mice some kind of f

Causes of Diabetes: Insulin, Mitochondria, Genetics, Metabolism | Scott Soleimanpour | 213
Short Summary: The science of diabetes with Dr. Scott Soleimanpour, blending personal insights and cutting-edge research that gets into mitochondrial biology and more.About the guest: Scott Soleimanpour, MD, is a physician-scientist at the University of Michigan’s Caswell Diabetes Institute, where he runs a lab studying pancreatic beta cell biology and mitochondrial quality control in diabetes.Note: Podcast episodes are fully available to paid subscribers on the M&M Substack and everyone on YouTube. Partial versions are available elsewhere. Full transcript and other information on Substack.Episode Summary: Nick Jikomes interviews Dr. Scott Soleimanpour about the biology of diabetes, exploring the pancreas’s dual role in digestion and blood sugar regulation via beta cells, which produce insulin. They discuss type 1 diabetes as an autoimmune condition with rising incidence and complex beta cell issues, and type 2 as a progressive disease tied to beta cell failure, not just obesity. The conversation covers mitochondrial function in beta cells, lifestyle factors like diet, and the limits of current treatments, offering practical advice for prevention and management.Key Takeaways:* The pancreas has two parts: the exocrine (98%) makes digestive enzymes, while the endocrine (1-2%) regulates blood sugar with hormones like insulin from beta cells.* Type 1 diabetes involves autoimmunity and intrinsic beta cell defects, with rates increasing due to environmental factors, not just genetics.* Type 2 diabetes hinges on beta cell dysfunction, not obesity alone—only 1 in 5 overweight people develop it, showing other factors matter.* Beta cells are energy-intensive and long-lived, relying on mitochondria; stress from overwork (e.g., excess insulin demand) can lead to failure.* Current diabetes drugs don’t rejuvenate beta cells; only TZDs (rarely used) preserve function, highlighting a gap in treatment.Related episode:* M&M #140: Obesogens, Oxidative Stress, Dietary Sugars & Fats, Statins, Diabetes & the True Causes of Metabolic Dysfunction & Chronic Disease | Robert Lustig*Not medical advice.* Full audio version: [Apple] [Spotify] [Elsewhere]* Full video version: [YouTube] [Rumble]* Support M&M if you find value in this content.* Episode transcript below.Episode Chapters:00:00:00 Intro00:05:37 Pancreas and Beta Cell Basics00:11:42 Scott’s Personal Journey with Type 1 Diabetes00:16:24 Pathogenesis of Type 1 Diabetes00:21:47 Rising Rates of Type 1 Diabetes00:27:06 Type 1 Treatment and Beta Cell Regeneration00:33:21 Type 2 Diabetes & Beta Cell Failure00:39:47 Insulin Resistance vs. Secretion in Type 200:47:03 Diet, Lifestyle, and Type 2 Diabetes00:55:50 Mitochondrial Biology in Beta Cells01:01:20 Pre-Diabetes to Diabetes Transition01:07:35 Reversibility of Type 2 Diabetes01:14:39 Intrinsic Beta Cell Problems in Type 201:26:44 Final Thoughts and Treatment GapsFull AI-generated transcript below. Beware of typos & mistranslations!Scott Soleimanpour 1:32 yeah. So, yeah, I run a pancreatic beta cell biology laboratory at the University of Michigan. I've been here for a little over 11 years. We're really interested in the regulation of mitochondrial quality control in beta cells and how that relates to the pathogenesis of both type one and type two diabetes. And then we try to use our basic laboratory to try to bridge questions that are relevant for understanding the pathogenesis of diabetes and and to hopefully learn insights that can be valuable for translation to the clinicNick Jikomes 2:19 and these pancreatic beta cells are obviously key for understanding diabetes. So let's just give everyone a very, very quick, very basic crash course in the pancreas and the beta cells in particular. What is the pancreas basically doing as an organ, and how are beta cells working at a very basic level in a normal, healthy individual? Yes, great question.Scott Soleimanpour 2:41 So I like to think of the pancreas is actually two organs together. So you have 98% of the pancreas. That is what we call the exocrine pancreas. That's the parts of the pancreas that are responsible for making digestive enzymes that will be fed into the gut to help you digest food, like fats, especially in your food. And then there's a whole, you know, inner interconnected series of ducks that collect that those digestive enzymes and then get them into the intestine. And then one to 2% of the pancreas are called the endocrine pancreas. That's what we study in my lab. And the endocrine pancreas is primarily organized into these structures called islets of longer Hans. And they're essentially micro organs that are made up of many different cell types that are kind of sprinkled throughout the pancreas, and the different cell types in the islet release hormones that have a lot to do with the regulation of glucose levels in the body. You mentioned the beta cells, the beta cells in a human comprise maybe 40 to 50% of the cells in the islet, and they make insulin, which is the

How Science Really Works: Meta-Research, Publishing, Reproducibility, Peer Review, Funding | John Ioannidis | 212
Short Summary: An insider’s look at the messy realities of scientific research with Stanford’s Dr. John Ioannidis. The good, the bad, and the ugly about how scientific research actually works.About the guest: John Ioannidis, MD, PhD is a professor at Stanford University in medicine, epidemiology, population health, and biomedical data science, with an MD from the University of Athens and a PhD from Harvard in biostatistics. He directs the Meta-Research Innovation Center at Stanford (METRICS), focusing on improving research methods and practices. Renowned for his paper “Why Most Published Research Findings Are False,” he’s among the most cited scientists globally, tackling biases and reproducibility in science.Note: Podcast episodes are fully available to paid subscribers on the M&M Substack and everyone on YouTube. Partial versions are available elsewhere. Full transcript and other information on Substack.Episode Summary: Nick Jikomes dives deep with John Ioannidis into the nuts and bolts of scientific research, exploring the replication crisis, the flaws of peer review, and the $30 billion publishing industry’s profit-driven quirks. They unpack Ioannidis’s controversial COVID-19 infection fatality rate estimates, the politicization of science, and the gaming of metrics like publication counts. The chat also covers NIH funding woes, administrative bloat, and Ioannidis’s current work on bettering research through transparency and new metrics.Key Takeaways:* Science’s “replication crisis” isn’t new—it’s baked into how tough and bias-prone research is, hitting all fields, not just “soft” ones like psychology.* Ioannidis’s famous claim, “most published findings are false,” holds up: stats show many “significant” results are flukes due to weak studies or bias.* Peer review’s a mixed bag—only a third of papers improve, and unpaid, tired reviewers miss a lot, letting shaky stuff slip through.* Publishing’s a $30 billion game with 50,000+ journals; big players like Elsevier rake in huge profits from subscriptions and fees, often over $10,000 per paper.* Researchers game the system—think fake co-authorships or citation cartels—boosting metrics like the H-index, which tracks papers with matching citation counts.* Ioannidis’s early COVID-19 fatality rate (0.2-0.3%) was spot-on but sparked a firestorm as politics warped science into “clan warfare.”* NIH funding’s clogged by red tape and favors older researchers, starving young innovators and risky ideas that could shake things up.* He’s building tools like a public database of scientist stats (4 million downloads!) to spotlight gaming and push for transparent, fair research.Related episode:* M&M #100: Infectious Disease, Epidemiology, Pandemics, Health Policy, COVID, Politicization of Science | Jay Bhattacharya*Not medical advice.* Full audio version: [Apple] [Spotify] [Elsewhere]* Full video version: [YouTube] [Rumble]* Support M&M if you find value in this content.* Episode transcript below.Episode Chapters:00:00:00 Intro 00:06:21 The Replication Crisis Explained 00:13:12 Replication in Science: How Much and Why? 00:18:14 Why Most Published Research Findings Are False 00:25:13 Peer Review: Strengths and Weaknesses 00:33:07 The Explosion of Journals and Predatory Publishing 00:41:40 The Business of Scientific Publishing 00:48:45 Open Access Costs and the Funding Dilemma 00:57:00 Preprints & Potential Solutions 01:04:34 Gaming the System: Metrics and Misconduct 01:11:08 COVID-19 & Politicization of Science 01:18:31 Revisiting the Infection Fatality Rate 01:25:48 NIH Funding & Leadership Changes 01:32:13 Directs vs. Indirects in Research Grants 01:40:56 Hopes for NIH Reform with Jay Bhattacharya 01:46:37 Current Projects & Closing ThoughtsFull AI-generated transcript below. Beware of typos & mistranslations!John Ioannidis 1:53 I'm a professor at Stanford in the Department of Medicine, of epidemiology and population health and biomedical data science. I'm running the meta research innovation center, or metrics at Stanford, which is a center focused on studying research and its processes, its practices, how we can make research methods and practices better. And I've worked in different fields, in evidence based medicine and other areas that it's very common to see problems with methods, with biases, with making errors, including prominently my own, I guess, and trying to be sensitized by them and try to see how we can improve efficiency andNick Jikomes 1:16 To find all of my content. Foreign.Thank you for joining me.John Ioannidis 2:39 eventually get the most out of this fascinating enterprise that we call science.Nick Jikomes 2:43 Yeah, and it really is an enterprise. There's lots of parts to the scientific research process. So you you've done a lot of work studying the scientific research process itself. You research research.John Ioannidis 2:57 It is research on research, and sometimes research on research and research. So there's no end to the method transformation.Ni