
Channel Your Enthusiasm
Channel Your Enthusiasm · joel topf
Show overview
Channel Your Enthusiasm has been publishing since 2021, and across the 5 years since has built a catalogue of 30 episodes. That works out to roughly 45 hours of audio in total. Releases follow a roughly quarterly cadence.
Episodes typically run over ninety minutes — most land between 1h 23m and 1h 45m — and the run-time is fairly consistent across the catalogue. None of the episodes are flagged explicit by the publisher. It is catalogued as a EN-language Health & Fitness show.
The show is actively publishing — the most recent episode landed 2 months ago, with 2 episodes already out so far this year. Published by joel topf.
From the publisher
A chapter by chapter recap of Burton Rose’s classic, The Clinical Physiology of Acid Base and Electrolyte Disorders, a kidney physiology book for nephrologists, fellows, residents and medical students.
Latest Episodes
View all 30 episodes
S1 Ep 31Chapter Twenty One: Respiratory Alkalosis
ReferencesChapter 19, Part 3 August 30, 2023Biff Palmer’s Ted Talk-Why not? Biff Palmer at TEDxSMU 2013Anna mentioned this issue of lactic acidosis in a panic disorder: The Lactic Acid Response to Alkalosis in Panic Disorder | The Journal of Neuropsychiatry and Clinical NeurosciencesReminder of important clinical lesson: Lactate: panicking doctor or panicking patient? - PMCMelanie regaled the group with an excerpt (page 351) Cohen, J. J., Kassirer, J. P. (1982). Acid-base. United States: Little, Brown.Biff Palmer! Respiratory Acidosis and Respiratory Alkalosis: Core Curriculum 2023Melanie loves this study of chronic respiratory alkalosis on participants to traveled to the High ALpine research station on the Jungfraujoch in the Swiss Alps Chronic Respiratory Alkalosis — The Effect of Sustained Hyperventilation on Renal Regulation of Acid–Base Equilibrium | NEJM (and here’s a great picture: Services: Jungfraujoch Research Station - Climate and Environmental Physics (CEP)JC mentioned that there are cells in the carotid body which are called glomus cells Neurobiology of the carotid body.JC discussed respiratory alkalosis in cirrhosis and here’s a review he had melanie write that addresses this topic: Acid Base Disorders in Cirrhosis - Advances in Kidney Disease and Health and here are some reviews he likes: The hyperventilation of cirrhosis: progesterone and estradiol effects and Acid-base disturbance in patients with cirrhosis: relation to hemodynamic dysfunction and Blood-Brain Barrier Permeability Is Exacerbated in Experimental Model of Hepatic Encephalopathy via MMP-9 Activation and Downregulation of Tight Junction ProteinsThe finding of respiratory alkalosis in pregnancy is not a new concept. Here’s a study from 1962: Acid-base balance of arterial blood during pregnancy, at delivery, and in the puerperium - American Journal of Obstetrics & GynecologyMelanie reminded us of the Charlie Brown sad face that occurs after bicarbonate infusion and delay in bicarbonate movement to the CSF! Spinal-Fluid pH and Neurologic Symptoms in Systemic Acidosis | NEJM (part 2 of chapter 11)Josh mentioned this report from Andrew Tarulli (a great neurologist previously at BIDMC who has moved to Overlook Hospital in NJ) Central Neurogenic Hyperventilation: A Case Report and Discussion of Pathophysiology | Allergy and Clinical Immunology | JAMA NeurologyHe also mentioned this important transporters that affect the pH. The choroid plexus sodium-bicarbonate cotransporter NBCe2 regulates mouse cerebrospinal fluid pHRefractory Central Neurogenic Hyperventilation: A Novel Approach Utilizing Mechanical Dead SpaceOutline: Chapter 21Respiratory AlkalosisIncreased pH, low pCO2, variable reduction in HCO3Differentiate from metabolic acidosis where pH is decreased(but pCO2 and HCO3 are likewise decreased)PATHOPHYSIOLOGYPrimary decrease in pCO2 when effective alveolar ventilation is increased beyond that needed to eliminate daily CO2 productionHow does the body respond to hypocapniaMass actionReduction in H+ induced by hypocapnia can be minimized by lowering HCO3One: rapid cell bufferingTwo: later decrease in net renal acid secretion → lower HCO3These two strategies explain the difference between acute and chronic respiratory alkalosisAcute Respiratory AlkalosisWithin 10 minutes, H ions move into extracellular fluidH+ combines with HCO3 → fall in plasma HCO3Converted to CO2 and H2OH+ comes from intracellular buffersProtein, phosphate, hemoglobinH+ may also come from alkalemia-induced increase in cellular lactic acid production (1)⁉️Enough H+ enters ECF to lower HCO3 by 2 mEq for each 10 mmHg decrease in pCO2 (Fig 20-3)Example: pCO2 falls to 20HCO3 falls by 4 → ~20 mEq/LpH ~7.63Not very efficient at protecting pHWithout compensation pH would be ~7.70Chronic Respiratory AlkalosisCompensatory ↓ renal H secretionBegins within 2 hoursNot complete for 2–3 daysDue to parallel rise in tubular cell pHManifested byHCO3 lossDecreased NH4 in urine4 mEq drop in HCO3 for each 10 mmHg decrease in pCO2Example: pCO2 20 → HCO3 16 → pH ~7.53ETIOLOGYRespiration governed by two sets of chemoreceptorsCentral (respiratory center in brainstem)Peripheral (carotid bodies at bifurcation, aortic bodies at arch)Central chemoreceptorsStimulated by ↑ pCO2 or metabolic acidosisPeripheral chemoreceptorsStimulated by hypoxia (and acidosis)Thus hyperventilation can be produced byHypoxemiaAnemiaReduction in arterial pHOther stimuliPainAnxietyMechanoreceptorsDirect stimulation of respiratory centerTable 21-1HypoxemiaRespiratory response occurs in stagesStage 1Peripheral chemoreceptor activationHyperventilation → respiratory alkalosisIncreased cerebral pH inhibits central respiratory centerLimits hyperventilationNo significant hyperventilation until pO2 < 50–60 mmHgIf lung disease prevents pCO2 reductionHypoxia stimulates ventilation at PaO2 < 70–80 mmHgStage 2⁉️Persistent hypoxemia → ↓ HCO3Lowers pH toward normalRemoves alkalosis inhibitionAllows greater ventilatory r

S1 Ep 30Chapter Nineteen: Metabolic Acidosis, part 3
ReferencesChapter 19, Part 3 August 30, 2023Joel and Roger mentioned the most common cause seems to be Sjögren’s syndrome for an acquired distal RTA. We mentioned this in an earlier episode and referenced this example of an absence of the H+ ATPase, presumably from autoantibodies to this transporter. Here’s a case report: Absence of H(+)-ATPase in cortical collecting tubules of a patient with Sjogren's syndrome and distal renal tubular acidosis Joel mentioned this paper in the New England Journal of Medicine in which there were patients who had hyperkalemia with a distal RTA: Hyperkalemic Distal Renal Tubular Acidosis Associated with Obstructive Uropathy | NEJM in this setting, some patients Anna mentioned this article on “ampho-terrible:” It’s the holes!!! Yano T, Itoh Y, Kawamura E, Maeda A, Egashira N, Nishida M, Kurose H, Oishi R. Amphotericin B-induced renal tubular cell injury is mediated by Na+ Influx through ion-permeable pores and subsequent activation of mitogen-activated protein kinases and elevation of intracellular Ca2+ concentration. Antimicrob Agents Chemother. 2009 Apr;53(4):1420-6Josh mentioned this study on furosemide’s effect on the TAL: Furosemide-induced urinary acidification is caused by pronounced H+ secretion in the thick ascending limb Urinary acidification assessed by simultaneous furosemide and fludrocortisone treatment: an alternative to ammonium chloride - Kidney InternationalMelanie mentioned treatment of patients with cystinosis Expert guidance on the multidisciplinary management of cystinosis in adolescent and adult patients | Clinical Kidney Journal | Oxford AcademicAmy shared her observations regarding base supplements including Prevention of recurrent calcium stone formation with potassium citrate therapy in patients with distal renal tubular acidosis - PubMed and Dosage of potassium citrate in the correction of urinary abnormalities in pediatric distal renal tubular acidosis patients - PubMedRoger mentioned that he has had good luck with Moonstone Nutrition drinks alkali citrates for kidney healthWe referred to David Goldfarb’s teaching on kidney stones in patients with acidification defects: A Woman with Recurrent Calcium Phosphate Kidney Stones (we also referenced this in an earlier episode but this one is a fan favorite). Joel mentioned the concern of bone loss in distal RTA: Incomplete renal tubular acidosis in 'primary' osteoporosis and Abnormal distal renal tubular acidification in patients with low bone mass: prevalence and impact of alkali treatmentJC mentioned Ehlers-Danlos syndrome with renal tubular acidosis and medullary sponge kidneys. A report of a case and studies of renal acidification in other patients with the Ehlers-Danlos syndromeLety mentioned concerns of encrustation of stents in stone forming individuals Potassium Citrate as a Preventive Treatment for Double-J Stent Encrustation: A Randomized Clinical TrialJoel schooled us in toluene and the presentation which appears to be an RTA- https://journals.lww.com/JASN/Abstract/1991/02000/Glue_sniffing_and_distal_renal_tubular_acidosis_.3.aspxMelanie mentioned this work by Alan Yu’s lab on a mechanism of hypercalciuria Claudin-2 deficiency associates with hypercalciuria in mice and human kidney stone disease Furosemide/Fludrocortisone Test and Clinical Parameters to Diagnose Incomplete Distal Renal Tubular Acidosis in Kidney Stone Formers and an accompanying editorial by Goldfarb Refining Diagnostic Approaches in Nephrolithiasis: Incomplete Distal Renal Tubular AcidosisHere’s a nice piece on ifosfamide and phosphate from Josh New clues for nephrotoxicity induced by ifosfamide: preferential renal uptake via the human organic cation transporter 2Here’s this crazy piece on excessive bicarbonate - Gas production after reaction of sodium bicarbonate and hydrochloric acidJosh points out that the pH can be important for inotropy: An effect of pH upon epinephrine inotropic receptors in the turtle heartMel’s favorite from Halperin because of the pun: Renal tubular acidosis (RTA): recognize the ammonium defect and pHorget the urine pHAmy’s VOG on RTA and OsteoporosisKI Review on acidosis and bone health: Effects of acid on boneGuideline on congenital RTA: Distal renal tubular acidosis: ERKNet/ESPN clinical practice pointsAJKD article on acidosis and bone health: Serum Bicarbonate and Bone Mineral Density in US AdultsCitrate reversing CsA induced acidosis effects: Citrate reverses cyclosporin A-induced metabolic acidosis and bone resorption in ratsOutline: Chapter 19 Metabolic Acidosis part 3Renal Tubular AcidosisAcidosis from diminished net tubular acid secretionThree typesType 1 (Distal)Type 2 (Proximal)Type 4 (…)The acidosis of renal failure could be added to this groupBut NH4+ per nephron is normalThis is a problem of too few nephrons, not tubular acidosisNephrons able to maximally acidify the urineType 1 Distal RTADecrease in net H secretion in the collecting duc

S1 Ep 29Chapter Nineteen: Metabolic Acidosis, part 2
ReferencesChapter 19, Part 12Metabolic acidosis June 14, 2023ReferencesChapter 19, Part 2Roger mentioned MELAS syndrome MELAS syndrome: Clinical manifestations, pathogenesis, and treatment optionsJosh mentioned this blog on lactate- Understanding lactate in sepsis & Using it to our advantageWe discussed the Warburg effect The Warburg Effect: How Does it Benefit Cancer Cells? - PMC and here’s a case from skeleton key- Skeleton Key Group Case #28: Mysterious Acidosis in Cancer - Renal Fellow NetworkOtto Warburg won the Nobel Prize in Physiology and Medicine in 1931 for describing how animal tumors produce large quantities of lactic acid (Wikipedia)Joel calls it the Lactate saline reflex, but the accepted term of art is Lacto-Bolo reflex The origins of the Lacto-Bolo reflex: the mythology of lactate in sepsisBuffer agents do not reverse intramyocardial acidosis during cardiac resuscitation.Josh mentioned this article the BICAR-ICU Sodium bicarbonate therapy for patients with severe metabolic acidaemia in the intensive care unit (BICAR-ICU): a multicentre, open-label, randomised controlled, phase 3 trial - The LancetRoger shared 3 quotes to make the point that there has been little movement in our knowledge the past 40 years:Bicarbonate does not improve hemodynamics in critically ill patients who have lactic acidosis. A prospective, controlled clinical study from Cooper in the AnnalsLactic Acidosis and Bicarbonate Therapy | Annals of Internal Medicine from Robert HollanderLactic acidosis from Nick MadiasJosh mentioned the use of sodium bicarbonate for CKD Eubicarbonatemic Hydrogen Ion Retention and CKD Progression - Kidney Medicine (Madias) Bicarbonate therapy for prevention of chronic kidney disease progression (from Wesson), Sodium Bicarbonate Prescription and Extracellular Volume Increase: Real‐world Data Results from the AlcalUN StudyAmy’s VoG on metabolic acidosis/KDIGO guidelinesVery nice JASN review that describes the mechanisms of how metabolic acidosis leads to CKD progressionFirst description by THE Dr. Bright1930 Lancet description of benefit2009 RCT that the 2012 KDIGO guidelines sort of based their 2b recommendations off of2020 BiCARB Study2021 META AnalysisWe discussed methanol toxicity : Case Study: Methanol Poisoning from Adulterated Liquor | Food Safety, Acute methyl alcohol poisoning: a review based on experiences in an outbreak of 323 cases and josh poking at the osmolar gap: PulmCrit- Toxicology dogmalysis: the osmolal gap and shared these guidelines: METHANOL | extrip-workgroup and Roger loves this: Urine fluorescence using a Wood's lamp to detect the antifreeze additive sodium fluorescein: a qualitative adjunctive test in suspected ethylene glycol ingestionsFrom China to Panama, a Trail of Poisoned Medicine - The New York Times (diethylene glycol) . The Accidental Poison That Founded the Modern FDA - The AtlanticOutline: Chapter 19 Metabolic AcidosisEtiologies and DiagnosisLactic AcidosisPyruvate → lactate (LDH; NADH → NAD+)Normal production: 15–20 mmol/kg/dayMetabolized in liver/kidney → pyruvate → glucose or TCANormal lactate: 0.5–1.5 mmol/L; acidosis if > 4–5 mmol/LCauses:↑ production: hypoxia, redox imbalance, seizures, exercise↓ utilization: shock, hepatic hypoperfusionMalignancy, alcoholism, antiretroviralsD-lactic acidosisShort bowel/jejunal bypassGlucose → D-lactate (not metabolized by LDH)Symptoms: confusion, ataxia, slurred speechSpecial assay neededTx: bicarb, oral antibioticsTreatmentUnderlying causeBicarb controversial: may worsen intracellular acidosis, overshoot alkalosis, ↑ lactateTarget pH > 7.1; prefer mixed venous pH/pCO2Ketoacidosis (Chapter 25 elaborates)FFA → TG, CO2, H2O, ketones (acetoacetate, BHB)Requires:↑ lipolysis (↓ insulin)Hepatic preference for ketogenesisCauses:DKA (glucose > 400)Fasting ketosis (mild)Alcoholic ketoacidosisPoor intake + EtOH → ↓ gluconeogenesis, ↑ lipolysisMixed acid-base (vomiting, hepatic failure, NAGMA)Congenital organic acidemias, salicylatesDiagnosis:AG, osmolar gap (acetone, glycerol)Ketones: nitroprusside only detects acetone/acetoacetateBHB can be 90% of total (false negative)Captopril → false positiveTreatment:Insulin +/- glucoseRenal Failure↓ excretion of daily acid loadGFR < 40–50 → ↓ ammonium/TA excretionBone buffering stabilizes HCO3 at 12–20 mEq/LSecondary hyperparathyroidism helps with phosphate bufferingAlkali therapy controversial in adultsIngestionsSalicylatesSymptoms at >40–50 mg/dLEarly: respiratory alkalosis → Later: metabolic acidosisTreatment: bicarb, dialysis (>80 mg/dL or coma)MethanolMetabolized to formic acid → retinal toxicityOsmolar gap elevatedTx: bicarb, ethanol/fomepizole, dialysisEthylene glycol→ glycolic/oxalic acid → renal failureSame treatment + thiamine/pyridoxineOtherToluene, sulfur, chlorine gas, hyperalimentation (arginine, lysine)GI Bicarbonate LossDiarrhea, bile/pancreatic drainage → loss of alkaline fluidsUreterosigmoidostomy → Cl-/HCO3- exchange in colonChole

S1 Ep 27Chapter Eighteen: Metabolic Alkalosis, part 2
ReferencesPart 2, March 1, 2023The alkaline tide phenomenon in studies that measured both the alkaline tide and acid secretion, the bicarbonate accumulation increased in linear fashion with the acid secretion. Melanie thought this was first recognized in the 60’s but later found this manuscript from 1939 in JCI! ALKALINE TIDES - PMCMelanie mentioned this old study that explores the respiratory response of metabolic acidosis and finds it “incomplete” compared to expected. EVALUATION OF RESPIRATORY COMPENSATION IN METABOLIC ALKALOSIS and there’s another image in a review by Michael Emmett Figure 1. Metabolic Alkalosis: A Brief Pathophysiologic Review - PMC(here’s the image from JCI) The effect of changes in blood pH on the plasma total ammonia level - SurgeryThis is an interesting case that Melanie mentioned with the help of Stew Lecker Trust the Patient: An Unusual Case of Metabolic Alkalosis - PMCGot Calcium? Welcome to the Calcium-Alkali Syndrome : Journal of the American Society of Nephrology a favorite review of the “calcium alkali” syndrome- previously called milk alkali syndrome but now milk is not commonly part of the syndrome (as with Dr. Sippie). Lety mentioned this issue with a new contaminant of street drugs: Tranq Dope: Animal Sedative Mixed With Fentanyl Brings Fresh Horror to U.S. Drug ZonesHere are two references that illustrate how the urine pH changes over the course of the day. Circadian variation in urine pH and uric acid nephrolithiasis risk The diurnal variation in urine acidification differs between normal individuals and uric acid stone formers - PMCNotes for Melanie’s VOG on reference 47: Maladaptive renal response to secondary hypercapnia in chronic metabolic alkalosisFrom Biff Palmer Figure 4- Respiratory Acidosis and Respiratory Alkalosis: Core Curriculum 2023 - American Journal of Kidney DiseasesAnna’s VOG- GI composition of cats or somethingOutline: Chapter 18Metabolic AlkalosisElevation of arterial pH, increased plasma HCO3, and compensatory hypoventilationHigh HCO3 may be compensatory for respiratory acidosisHCO3 > 40 indicates metabolic alkalosisPathophysiology: Two Key QuestionsHow do patients become alkalotic?Why do they remain alkalotic?Generation of Metabolic AlkalosisLoss of H+ ionsGI loss: vomiting, GI suction, antacidsRenal loss: diuretics, mineralocorticoid excess, hypercalcemia, post-hypercapniaAdministration of bicarbonateTranscellular shiftK+ loss → H+ shifts intracellularlyIntracellular acidosisRefeeding syndromeContraction alkalosisSame HCO3, smaller extracellular volume → increased [HCO3]Seen in CF (sweating), illustrated in Fig 18-1Common theme: hypochloremia is essential for maintenanceMaintenance of Metabolic AlkalosisKidneys normally excrete excess HCO3Example: Normal subjects excrete 1000 mEq NaHCO3/day with minor pH changeImpaired HCO3 excretion required for maintenanceTable 18-2Mechanisms of MaintenanceDecreased GFR (less important)Increased tubular reabsorptionProximal tubule (PT): reabsorbs 90% of filtered HCO3TALH and distal nephron manage the restContributing factors:Effective circulating volume depletionEnhances HCO3 reabsorptionAng II increases Na-H exchangeIncreased tubular [HCO3] enables more H+ secretionDistal nephron HCO3 reabsorptionStimulated by aldosterone (↑ H-ATPase, ↑ Na reabsorption)Negative luminal charge impedes H+ back-diffusionChloride depletionReduces NaK2Cl activity → ↑ renin → ↑ aldosteroneLuminal H-ATPase co-secretes Cl → low Cl increases H+ secretionCl-HCO3 exchanger needs Cl gradient → low Cl impairs HCO3 secretionKey conclusion: Cl depletion > volume depletion in perpetuating alkalosisAlbumin corrects volume but not alkalosisNon-N Cl salts correct alkalosis without fixing volumeHypokalemiaStimulates H+ secretion and HCO3 reabsorptionTranscellular shift (H/K exchange) → intracellular acidosisH-K ATPase reabsorbs K and secretes HSevere hypokalemia reduces Cl reabsorption → ↑ H+ secretionImportant with mineralocorticoid excessRespiratory CompensationHypoventilation: 0.7 mmHg PCO2 ↑ per 1 mEq/L HCO3 ↑PCO2 can exceed 60Rise in PCO2 increases acid excretion (limited effect on pH)EpidemiologyGI Hydrogen LossGastric juice: high HCl, low KClStomach H+ generation → blood HCO3Normally recombine in duodenumVomiting/antacids prevent recombination → alkalosisAntacids (e.g., MgOH)Mg binds fats, leaves HCO3 unbound → alkalosisRenal failure impairs excretionCation exchange resins (SPS, MgCO3) → same effectCongenital chloridorrheaHigh fecal Cl-, low pH → metabolic alkalosisPPI may help by reducing gastric Cl loadRenal Hydrogen LossMineralocorticoid excess & hypokalemiaAldosterone → H+ ATPase stimulation, Na+ reabsorption → negative lumen → ↑ H+ secretionDiuretics (loop/thiazide)Volume contractionSecondary hyperaldosteronismIncreased distal flow and H+ lossPosthypercapnic alkalosisChronic respiratory acidosis → ↑ HCO3Rapid correction (ventilation) → unopposed HCO3 → alkalosisGradual CO2 correction neededMaintenance: hypox

S1 Ep 28Chapter Nineteen: Metabolic Acidosis, The Show, part 1
ReferencesChapter 19, Part 1 Metabolic acidosis June 14, 2023American Society of Nephrology | Medical Students - Kidney TREKS this is the program that Josh mentioned at Mount Desert Island! Effects of pH on Potassium: New Explanations for Old Observations - PMC here’s the review melanie from Peter Aronson that clarifies the fact that there are no H+-K+ antiporters outside the kidney but rather coupled transport-We discussed whether we like “Winter’s formula” Quantitative Displacement of Acid-Base Equilibrium in Metabolic Acidosis | Annals of Internal Medicine Dr. R. W. Winters was charged with larceny https://www.nytimes.com/1982/05/16/nyregion/ex-columbia-u-doctor-charged-with-larceny.htmlJCI - The Maladaptive Renal Response to Secondary Hypocapnia during Chronic HCl Acidosis in the Dog this was a classic experiment exploring the respiratory response to an infusion of HCl but the animals were maintained in a high pCO2 milieu (not generalizable to humans!)Here’s the thoughtful Pulmcrit post (by Josh Farkas) that Josh mentioned regarding correction of anion gap for hypoalbuminemia: Mythbusting: Correcting the anion gap for albumin is not helpfulJC mentioned that the anion gap does change in cirrhosis when the albumin is very low but using the correction factor may not change the clinical findings Acid-base disturbance in patients with cirrhosis: relation to hemodynamic dysfunctionDiagnostic Importance of an Increased Serum Anion Gap | NEJM Melanie mentioned the work of Patricia Gabow on the anion gap. In this review, she refers to work that she had done to try to identify all the organic anions in the anion gap but it falls short. Also, check out this critical look at the delta/delta: The Δ Anion Gap/Δ Bicarbonate Ratio in Lactic Acidosis: Time for a New Baseline?Roger mentioned near drowning in the Dead Sea and the unusual electrolytes in that instance. Near-Drowning in the Dead Sea: A Retrospective Observational Analysis of 69 PatientsWe discussed this classic NEJM article by Daniel Batlle The Use of the Urinary Anion Gap in the Diagnosis of Hyperchloremic Metabolic AcidosisAmy mentioned this review from Uribarri and Oh in JASN on the urine anion gap: The Urine Anion Gap: Common MisconceptionsJoel has a great blog post on the urine osmolar gap. urine osmolar gap – Precious Bodily Fluids Anna’s VoG on the bicarb deficit: Kurtz, I Acid-Base Case Studies, 2nd Edition. Trafford Publishing 2004. And the Fernandez paper that derived a better equationReference for Josh’s VoG: Key enzyme in charge of ketone reabsorption of renal tubular SMCT1 may be a new target in diabetic kidney diseaseSevere anion gap acidosis associated with intravenous sodium thiosulfate administrationUnexpectedly severe metabolic acidosis associated with sodium thiosulfate therapy in a patient with calcific uremic arteriolopathySodium Thiosulfate Induced Severe Anion Gap Metabolic AcidosisSodium Thiosulfate and the Anion Gap in Patients Treated by HemodialysisOutline: Chapter 19 Metabolic AcidosisOverviewLow arterial pHReduced HCO3Compensatory hyperventilation (↓ pCO2)Bicarb < 10 strongly suggests metabolic acidosis (renal compensation for respiratory alkalosis does not go that low)PathophysiologyH+ + HCO3- <=> H2CO3 <=> CO2 + H2OAcidosis results from H+ addition or HCO3 lossResponse to Acid LoadExtracellular bufferingExample: Add 12 mmol H+/L → HCO3 falls from 24 → 12 → pH drops to 7.1 (40 to 80 nmol/L)Intracellular and bone buffering55–60% buffered intracellularly and in bone12 mEq/L acid load only reduces serum HCO3 by ~5 mEq/LH+ into cells → K+ out (hyperkalemia)Notably in diarrhea or renal failureLess effect with organic acidosis (e.g., DKA, lactic acidosis)Respiratory compensationStimulates chemoreceptors → ↑ tidal volume (more than RR)Decreases pCO2, increases pHBegins within 1–2 hours; peaks at 12–24 hoursWinters formula alternative: for every 1 mEq ↓ HCO3, pCO2 ↓ by 1.2Chronic: respiratory compensation is blunted by renal adaptationRenal hydrogen excretion50–100 mEq/day acid generated from diet90% filtered HCO3 reabsorbed in PTAcid secreted:10–40 mEq via titratable acid (TA)30–60 mEq via NH3/NH4 (can ↑ to 250 mEq in acidosis)TA: phosphate (DKA → ketones act as TA)Max excretion up to 500 mEq/day in severe acidosisGeneration of Metabolic AcidosisMechanismsInability to excrete H+ (slow)Addition of H+ or loss of HCO3 (rapid)Anion Gap (AG)Normal: 5–11 (falling due to rising Cl-)Mostly due to negatively charged proteins (albumin)Adjust for albumin: AG ↓ 2.5 per 1 g/dL albumin ↓Revised: AG = unmeasured anions - unmeasured cations↑ AG = addition of unmeasured anions (e.g., lactate, ketones)Hyperchloremic acidosis: ↓ HCO3 replaced by ↑ Cl (normal AG)Delta–Delta AnalysisAdjust AG for albuminNormal ΔAG:ΔHCO3 = 1.6:1 (early 1:1)<1 → high + normal AG acidosisOther causes of AG variationHigh AG without acidosis: hemoconcentration, alkalosisLow AG: hypoalbuminemia, ↑ un

S1 Ep 26Chapter Eighteen: Metabolic Alkalosis, part 1
We are a bit slappy at the beginning of the episode since we had just recorded our conversation with the Glaucomfleckens. ReferencesChapter 18 Metabolic alkalosis! Part 1 February 23, 2023It is chloride depletion alkalosis, not contraction alkalosis classic review by Galla and Luke, the metabolic alkalosis mavens who review the role of chloride.On the mechanism by which chloride corrects metabolic alkalosis in man and this is the study when they induced a metabolic alkalosis and studied the effect of treating with KCl vs NaPhos and found the former (with chloride) reversed the alkalosis but not the sodium containing protocol. Some elegant reports on the increased proximal reabsorption of bicarbonate above normal stimulated by Ang II. Tubular transport responses to angiotensin | American Journal of Physiology-Renal PhysiologyCrosstalk between the renal sympathetic nerve and intrarenal angiotensin II modulates proximal tubular sodium reabsorption - Pontes - 2015 - Experimental Physiology - Wiley Online LibraryTHE RENAL REGULATION OF ACID-BASE BALANCE IN MAN. III. THE REABSORPTION AND EXCRETION OF BICARBONATE 1949 this is the correct figure for 11.14 and shows what happens when filtered bicarb exceeds normal threshold in normal human (men) and appears in the urine. Masterful review Symposium on acid-base homeostasis. The generation and maintenance of metabolic alkalosis by Seldin and Rector And a modern review from Michael Emmet! Metabolic Alkalosis - PMC (so many favorite reviews on this exciting topic!) and this one from Soleimani Metabolic Alkalosis Pathogenesis, Diagnosis, and Treatment: Core Curriculum 2022 both of these elaborate on pendrin’s role. The effect of prolonged administration of large doses of sodium bicarbonate in man (Clin Sci. 1954 Aug;13(3):383-401)Kidney v Renal: KDIGO versus Don’tPlus: We got a little off topic and discussed the Kidney Failure Risk Equation: https://kidneyfailurerisk.com/Outline: Chapter 18Metabolic AlkalosisElevation of arterial pH, increased plasma HCO3, and compensatory hypoventilationHigh HCO3 may be compensatory for respiratory acidosisHCO3 > 40 indicates metabolic alkalosisPathophysiology: Two Key QuestionsHow do patients become alkalotic?Why do they remain alkalotic?Generation of Metabolic AlkalosisLoss of H+ ionsGI loss: vomiting, GI suction, antacidsRenal loss: diuretics, mineralocorticoid excess, hypercalcemia, post-hypercapniaAdministration of bicarbonateTranscellular shiftK+ loss → H+ shifts intracellularlyIntracellular acidosisRefeeding syndromeContraction alkalosisSame HCO3, smaller extracellular volume → increased [HCO3]Seen in CF (sweating), illustrated in Fig 18-1Common theme: hypochloremia is essential for maintenanceMaintenance of Metabolic AlkalosisKidneys normally excrete excess HCO3Example: Normal subjects excrete 1000 mEq NaHCO3/day with minor pH changeImpaired HCO3 excretion required for maintenanceTable 18-2Mechanisms of MaintenanceDecreased GFR (less important)Increased tubular reabsorptionProximal tubule (PT): reabsorbs 90% of filtered HCO3TALH and distal nephron manage the restContributing factors:Effective circulating volume depletionEnhances HCO3 reabsorptionAng II increases Na-H exchangeIncreased tubular [HCO3] enables more H+ secretionDistal nephron HCO3 reabsorptionStimulated by aldosterone (↑ H-ATPase, ↑ Na reabsorption)Negative luminal charge impedes H+ back-diffusionChloride depletionReduces NaK2Cl activity → ↑ renin → ↑ aldosteroneLuminal H-ATPase co-secretes Cl → low Cl increases H+ secretionCl-HCO3 exchanger needs Cl gradient → low Cl impairs HCO3 secretionKey conclusion: Cl depletion > volume depletion in perpetuating alkalosisAlbumin corrects volume but not alkalosisNon-N Cl salts correct alkalosis without fixing volumeHypokalemiaStimulates H+ secretion and HCO3 reabsorptionTranscellular shift (H/K exchange) → intracellular acidosisH-K ATPase reabsorbs K and secretes HSevere hypokalemia reduces Cl reabsorption → ↑ H+ secretionImportant with mineralocorticoid excessRespiratory CompensationHypoventilation: 0.7 mmHg PCO2 ↑ per 1 mEq/L HCO3 ↑PCO2 can exceed 60Rise in PCO2 increases acid excretion (limited effect on pH)EpidemiologyGI Hydrogen LossGastric juice: high HCl, low KClStomach H+ generation → blood HCO3Normally recombine in duodenumVomiting/antacids prevent recombination → alkalosisAntacids (e.g., MgOH)Mg binds fats, leaves HCO3 unbound → alkalosisRenal failure impairs excretionCation exchange resins (SPS, MgCO3) → same effectCongenital chloridorrheaHigh fecal Cl-, low pH → metabolic alkalosisPPI may help by reducing gastric Cl loadRenal Hydrogen LossMineralocorticoid excess & hypokalemiaAldosterone → H+ ATPase stimulation, Na+ reabsorption → negative lumen → ↑ H+ secretionDiuretics (loop/thiazide)Volume contractionSecondary hyperaldosteronismIncreased distal flow and H+ lossPosthypercapnic alkalosisChronic respiratory acidosis → ↑ HCO3Rapid correction (ventilation) → unoppos

S1 Ep 25Chapter Seventeen: Introduction to Simple and Mixed Acid-Base Disorders
ReferencesI said I used MDCalc but I was mistaken I use MedCalX which is nice but getting dated. We talked about this out of print book that we love: Cohen, J. J., Kassirer, J. P. (1982). Acid-base. United States: Little, Brown.Josh mentioned this article that looked at over 17,000 samples with simultaneous measured and calculated bicarbonate and found a very small difference. Comparison of Measured and Calculated Bicarbonate Values | Clinical Chemistry | Oxford AcademicBase deficit or excess- Diagnostic Use of Base Excess in Acid–Base Disorders | NEJM (check out the accompanying letter to the editor from Melanie challenging this article! Along with colleagues Lecker and Zeidel Diagnostic Use of Base Excess in Acid-Base Disorders )Melanie loves this paper which shows a nice correlation between arterial and venous pH but the rest of the comparisons are disappointing - Comparison of arterial and venous pH, bicarbonate, Pco2 and Po2 in initial emergency department assessment - PMCA nomogram for the interpretation of acid-base data is figure 17-1 in the book, this manuscript with the ! in the conclusion creates the acid-base map. We debated about whether we like Winter’s formula: Quantitative displacement of acid-base equilibrium in metabolic acidosis (melanie does b/c it used real patients). Amy’s Voice of God on Dietary Acid LoadReview of dietary acid load: https://pubmed.ncbi.nlm.nih.gov/23439373/, https://pubmed.ncbi.nlm.nih.gov/38282081/, https://pubmed.ncbi.nlm.nih.gov/33075387/Survey data from kidney stone formers regarding sources of dietary acid load: https://pubmed.ncbi.nlm.nih.gov/35752401/Urine profile for vegans and omnivories (urine pH and cations/anions): https://pubmed.ncbi.nlm.nih.gov/36364731/SWAP-MEAT pilot trial: https://pubmed.ncbi.nlm.nih.gov/39514692/ looked at urine profile on plant based meat diet (Beyond Meat) versus animal based meat dietNot all plant meat substitutes are the same in terms of net acid load: https://pubmed.ncbi.nlm.nih.gov/38504022/Frassetto paper showing that the dietary acid load effect is mostly from sodium chloride: https://pubmed.ncbi.nlm.nih.gov/17522265/Healthy eating is probably more important than plant based diet for CKD: https://pubmed.ncbi.nlm.nih.gov/37648119/, https://pubmed.ncbi.nlm.nih.gov/32268544/KDIGO 2024 guidelines: https://kdigo.org/guidelines/ckd-evaluation-and-management/Association (or lack thereof) of a pro-vegetarian diet and sarcopenia/protein energy wasting in CKD: https://pubmed.ncbi.nlm.nih.gov/39085942/Outline Chapter 17 Introduction to simple and mixed acid-base disordersIntroduction to Simple and Mixed Acid-Base DisordersDisturbances of acid-base homeostasis are common clinical problemsDiscussed in Chapters 18-21This chapter reviews:Basic principles of acid-base physiologyMechanisms of abnormalitiesEvaluation of simple and mixed acid-base disordersAcid-Base PhysiologyFree hydrogen is maintained at a very low concentration40 nanoEq/L1 millionth the concentration of Na, K, Cl, HCO3H+ is highly reactive and must be kept at low concentrationsCompatible H concentration: 16 to 160 nanoEq/LpH range: 7.8 to 6.8Buffers prevent excessive variation in H concentrationMost important buffer: HCO3Reaction: H+ + HCO3 <=> H2CO3 <=> H2O + CO2H2CO3 exists at low concentration compared to its productsHenderson-Hasselbalch Equation (HH Equation)Understanding acid-base can use both H+ concentration and pHMeasurement of pHMust be measured anaerobically to prevent CO2 lossMeasurement methods:pH: Electrode permeable to H+PCO2: CO2 electrodeHCO3: Calculated using HH EquationAlternative: Add strong acid, measure CO2 releasedPCO2 * 0.03 gives mEq of CO2Measured vs. Calculated HCO3pKa of 6.1 and PCO2 coefficient (0.03) varyMeasurement of CO2 prone to errorDebate remains unresolvedDifferences affect anion gap calculationsArterial vs. Venous Blood Gas (ABG vs. VBG)Venous pH is lower due to CO2 retentionVenous blood may be as accurate as arterial for pH if well perfusedPitfalls in pH MeasurementMust cool ABG quickly to prevent glycolysisAir bubbles affect gas readingsHeparin contamination lowers pHArterial pH may not reflect tissue pHReduced pulmonary blood flow skews resultsEnd tidal CO2 > 1.5% indicates adequate venous returnRegulation of Hydrogen ConcentrationHCO3/CO2 as the Principal BufferHigh HCO3 concentrationIndependent regulation of HCO3 (renal) and PCO2 (lungs)Renal Regulation of HCO3H secretion reabsorbs filtered bicarbonateLoss of HCO3 in urine equates to H retentionH combines with NH3 or HPO4, forming new HCO3Pulmonary Regulation of CO2CO2 is not an acid but forms H2CO3Lungs excrete 15,000 mmol of CO2 dailyKidneys excrete 50-100 mmol of H dailyH = 24 * (PCO2 / HCO3)pH compensation via respiratory and renal adjustmentsAcid-Base DisordersDefinitionsAcidemia: Decreased blood pHAlkalemia: Increased blood pHAcidosis: Process lowering pHAlkalosis: Process raising pHPrimary PCO2 abnormalities: Respiratory disordersPrimary HCO3 abno

S1 Ep 24Chapter Sixteen: Edematous States, part 2
ReferencesWe talked about winning the 2022 ASN innovation contest and here’s a link to our promo video https://www.dropbox.com/scl/fi/g4osnf0nradsfryyo51fi/ASN-Education-Contest-Channel-Your-Enthusiasm-Podcast.mp4?rlkey=pnso45x07nr3pane9y8cux8yg&e=1&dl=0We wondered about “permissive hypercreatinemia” and Josh referenced the DOSE trial: Relevance of Changes in Serum Creatinine During a Heart Failure Trial of Decongestive Strategies: Insights From the DOSE Trial - PMCPlus this editorial by Steve Coca: Ptolemy and Copernicus Revisited: The Complex Interplay between the Kidneys and Heart FailureWe refer to the Frank-Starling curve and reference an image from this paper by Jay Cohen: Blood pressure and cardiac performance - ScienceDirectWe felt that this chapter is dated with respect to heart failure. Check out this 2022 AHA/ACC/HFSA Guideline for the Management of Heart Failure: A Report of the American College of Cardiology/American Heart Association Joint Committee on Clinical Practice GuidelinesUnderfilling versus overflow in hepatic ascites an editorial by Frank Epstein Effect of Head-Out Water Immersion on Hepatorenal Syndrome - American Journal of Kidney Diseases studies done by Schrier which Roger mentionedThe fading concept: https://www.tandfonline.com/doi/abs/10.3109/00365528309182102?journalCode=igas2Historical Aspects of Ascites and the Hepatorenal Syndrome - Wong - 2021 - Clinical Liver Disease - Wiley Online LibraryHere’s a great paper from Andrew Allegretti on HRS prognosis: Prognosis of Patients with Cirrhosis and AKI Who Initiate RRT - PubMedJoel mentions landmark paper in NEJM for treating SBP Effect of Intravenous Albumin on Renal Impairment and Mortality in Patients with Cirrhosis and Spontaneous Bacterial Peritonitis | New England Journal of MedicineAlbumin infusion in patients undergoing large‐volume paracentesis: A meta‐analysis of randomized trials - Bernardi - 2012 - Hepatology - Wiley Online LibraryJoel wondered about the lore that minoxidil could lead to renal recovery: Minoxidil treatment of malignant hypertension. Recovery of renal functionRoger recalled an agent diazoxide: Hyperstat - Side Effects, Uses, Dosage, Overdose, Pregnancy, Alcohol | RxWikiHere’s an entertaining review on whether insulin leads to sodium retention: Invited Review: Sodium-retaining effect of insulin in diabetes - PMCInvasive monitoring for hemodynamicsFACTT: https://www.nejm.org/doi/full/10.1056/NEJMoa062200ESCAPE: https://pubmed.ncbi.nlm.nih.gov/16204662/PACMAN: https://pubmed.ncbi.nlm.nih.gov/16084255/EVEREST trial and use of tolvaptan in HFrEFEVEREST: https://pubmed.ncbi.nlm.nih.gov/17384437/Post-hoc analysis of hyponatremic patients of EVEREST: https://pubmed.ncbi.nlm.nih.gov/23743487/Outline Chapter 16 — Edematous States part 2Symptoms and diagnosisThree factors important in the mechanism of edemaThe pattern of distribution of edema which reflects those capillaries with altered hemodynamic forcesThe central venous pressurePresence or absence of pulmonary edemaPulmonary edemaShortness of breath and orthopneaTachypnic, diaphoretic, wet rales, gallops, murmursCheck a chest x-rayCardiac disease is most commonBut differential includes primary renal Na retention and ARDSWedge pressure will exceed 18-20 mmHg with heart or primary Na retention, but is relatively normal with ARDSUncomplicated cirrhosis does not cause pulmonary edemaIncreased capillary pressure in this disorder is only seen below the hepatic veinNormal or reduced blood volume in the cardiopulmonary circulationPeripheral edema and ascitesPeripheral edema is cosmetically undesireable but produces less serious symptomsSymptoms: swollen legs, difficulty walking, increased abdominal girth, shortness of breath due to pressure on the diaphragm.Pitting edema found in dependent areasAscites found in abdomenNephrotic syndrome low tissue pressure areas like eye orbitsHeart Failure (right sided) peripheral edema, abdominal wall, SOB is due to concomitant pulmonary disease. Right sided heart failure increases venous pressureCirrhosis develop cirrhosis and lower extremity edema, pressure above the hepatic vein is normal or low.Tense ascites can increase the pressure above the diaphragm but is relieved with a tapPortal pressure > 12 mmHg required for fluid retentionLove the case history 16-1Primary renal sodium retentionPulmonary and peripheral edemaJugular venous pressure is elevatedNephrotic SyndromePeriorbital and peripheral edema, rarely ascitesCVP normal to highIdiopathic edemaBehaves as volume depleted (especially with diuretics)Etiology and treatmentGeneral principles of treatmentWhen must edema be treatedWhat are the consequences of the removal of fluidHow rapidly should fluid be removedWhenPulmonary edema is the only form of generalized edema that is life threatening and demands immediate treatmentImportant for note: laryngeal edema and angioedema. Cerebral edemaWhat are the consequencesIf the edema fluid is compensatory (heart failure,

S1 Ep 23Chapter Sixteen: Edematous States, part 1
ReferencesCapillary Hemodynamics Insights into Salt Handling and Blood Pressure | NEJMAmy mentioned about the 3 phases of the interstitiumAre the precapillary sphincters and metarterioles universal components of the microcirculation? An historical review - PMCSafety factor?Renal Function during Recovery from Minimal Lesions Nephrotic Syndrome - Abstract - Nephron 1987, Vol. 47, No. 3 - Karger PublishersAre diuretics effective for idiopathic lymphedema? : Evidence-Based PracticeRapid diuresis in patients with ascites from chronic liver disease: the importance of peripheral edema for fig 16-7Activation and Inhibition of Sodium-Hydrogen Exchanger Is a Mechanism That Links the Pathophysiology and Treatment of Diabetes Mellitus With That of Heart FailureAdditional notes from our chat (might be overlap with Amy’s notes belowNew insights into the pathophysiology of edema in nephrotic syndrome by Helbert RondonThe hyperlipidemia of the nephrotic syndrome. Relation to plasma albumin concentration, oncotic pressure, and viscosityPlasmin in Nephrotic Urine Activates the Epithelial Sodium ChannelLipoprotein metabolism in experimental nephrosisViscosity regulates apolipoprotein A-1 gene expression in experimental models of secondary hyperlipidemia and in cultured hepatocytesAmiloride in Nephrotic Syndrome | Clinical Research Trial Listing ( oedema | EdemaHypoalbuminemia and proteinuria contribute separately to reduced lipoprotein catabolism in the nephrotic syndromeOrigin of hypercholesterolemia in chronic experimental nephrotic syndromeExtrahepatic lipogenesis contributes to hyperlipidemia in the analbuminemic ratApolipoprotein gene expression in analbuminemic rats and in rats with Heymann nephritisAmy’s NotesJosh “Blessed are the days” https://link.springer.com/article/10.1007/s00467-013-2435-6Amy mentions mels’ article Capillary Hemodynamics Insights into Salt Handling and Blood Pressure | NEJM, the 3 phases of the interstitiumJosh mentions a re: management of idiopathic edema (from up to date: https://www.uptodate.com/contents/idiopathic-edema)Amy stemmer sign: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6635205/, https://pubmed.ncbi.nlm.nih.gov/31281100/Anna in chat talking about amiloride, ENaC re: edema: https://www.researchgate.net/publication/50989884_New_insights_into_the_pathophysiology_of_edema_in_nephrotic_syndromeOutline Chapter 16 — Edematous StatesEdema is a palpable swelling produced by expansion of the interstitial fluid volumeConditions associated with this Heart failure Cirrhosis Nephrotic syndromePathophysiology of edema formationTwo steps Alteration of capillary hemodynamics that favors movement of fluid out of the capillary Dietary sodium and water are retained by the kidneyEdema does not become clinically apparent until interstitial volume has increased 2.5 to 3 liters If this fluid came the plasma would have hemoconcentration and shock Instead as fluid moves from vascular space to interstitium you get decreased tissue perfusion leading to kidney Na and water retention Net result is expansion of total extracellular volume with maintenance of plasma volume at close to normal levels This means that the kidney is responding appropriately.Important because therapy with diuretics will break this response and may diminish tissue perfusion. There are other situations where primary abnormality is inappropriate renal fluid retention. Here both the plasma and interstitial volumes are expanded and there is no consequences from diuretic therapy. This is over filling. Seen in cirrhosis, primary renal disease. Certain drugs Capillary hemodynamics Exchange of fluids at teh capillary is determined by the hydraulic and oncotic pressures in each compartment This can be expressed by Starlings law Net filtration = LpS (delta hydraulic pressure - delta oncotic pressure) Lp is the unit permeability or porosity of the capillary wall. S is the surface area. Sigma is the reflection coefficient ranging from zero for completely permeable to 1 for for impermeable Difficult to measure these values in humans and animals 16-1 is a table of starling force values. No reflection coefficient though Figure 16-2 shows values in subcutaneous tissues. PCap 17.3 Oncotic pressure in cap is 28. Says mean net gradient is 0.3 mmHg favoring filtration out of the vascular space. This excess net is returned to the systemic circulation by lymphatics. In the liver the values are different. The hepatic sinusoids are highly permeable to protein so oncotic pressure is neutralized by zero reflection coefficient. SO hydraulic pressure favoring filtration is unopposed. Cap hydraulic pressure is lower since two thirds of hepatic blood flow is from low pressure portal vein. Still large pressure gradient favoring filtrationAlveolar capillaries are similar to the liver Low cap hydraulic pressure, more permeable to proteins so no transcapillary oncotic pressure. Edema formation requires alteration of one or more starling forces to favor net f

S1 Ep 22Chapter Fifteen: Clinical Use of Diuretics, part 2
ReferencesProximal Tubule-Specific Deletion of the NHE3 (Na+/H+ Exchanger 3) in the Kidney Attenuates Ang II (Angiotensin II)-Induced Hypertension in Mice Melanie is in love with this paper that shows that sodium retention Bumetanide and furosemide in heart failure everyone agreed that we love this classic paper from Craig Brater on diuretics (and the source of figure 15-6). Lety referenced the Cr x 20 formula, a strategy to multiply the serum creatinine by 20 to estimate the initial furosemide dose. We agreed that this is more appropriate than the House of God formula of age + BUN = dose (which may be so much higher). Joel shared this excellent report Diuretic Optimization Strategies Evaluation (DOSE) trial: https://www.nejm.org/doi/full/10.1056/nejmoa1005419Amy shared how much she likes the two hour urine sodium (or random urine sodium) Rapid and Highly Accurate Prediction of Poor Loop Diuretic Natriuretic Response in Patients With Heart Failure - PMCAnna shared this paper which suggests that urinary sodium is more closely linked to outcome compared to urine volume Natriuretic Response Is Highly Variable and Associated With 6-Month Survival: Insights From the ROSE-AHF Trial and the study showing Substantial Discrepancy Between Fluid and Weight Loss During Acute Decompensated Heart Failure Treatment Josh worried about obstructive sleep apnea and nocturia: Sleep disordered breathing and nocturnal polyuria: nocturia and enuresis.WAITING FOR JOSHJC mentioned this report from a group in the Netherlands regarding solute load and urine volume Determinants of Urine Volume in ADPKD Patients Using the Vasopressin V2 Receptor Antagonist TolvaptanWe also considered CLICK trail Chlorthalidone for Hypertension in Advanced Chronic Kidney Disease | NEJM (and here’s the Freely Filtered Podcast on this topic- a really great episode! Freely Filtered 040: Double CLICK for BP control in CKD stage 4 — NephJCRoger shared these articles on albumin and furosemide: Co-administration of albumin-furosemide in patients with the nephrotic syndrome and Albumin and Furosemide Combination for Management of Edema in Nephrotic Syndrome: A Review of Clinical Studies - PMC. This is an interesting study that showed that the serum and urine albumin does not predict of the response to loop diuretics.Serum and Urine Albumin and Response to Loop Diuretics in Heart Failure | American Society of Nephrology JC”s abstract on use of loop diuretics in hepatorenal syndrome type 1 was ultimately published in the American Journal of the Medical Sciences: https://doi.org/10.1016/S0002-9629(23)00623-7Defining the role of albumin infusion in cirrhosis-associated hyponatremia this article explores the Gibbs-Donan Effect that Amy loves teaching us about. Distal Convoluted Tubule | American Society of Nephrology Figure 1 is a favorite (and a prerequisite to friendship with melanie)There is also a nice discussion of diuretic resistance in this year’s Nephmadness #NephMadness 2022: Cardiorenal Region – AJKD BlogJosh is excited about starting an SGLT2 inhibitor for acute heart failure and Anna mentions this article about how they may prevent AKI: The SGLT2 Inhibitor Empagliflozin Might Be a New Approach for the Prevention of Acute Kidney InjuryJosh remembered this Tweetorial from Avi Cooper on the direct effect of furosemide: https://twitter.com/avrahamcoopermd/status/1292134482812604418?lang=enRoger reminded us about the practice of using bedrest for heart failure: Prolonged Bed Rest in the Treatment of the Dilated Heart and rotating tourniquets Effectiveness of Congesting Cuffs ("Rotating Tourniquets") in Patients with Left Heart Failure | Circulation and Rotating Tourniquets for Acute Cardiogenic Pulmonary Edema | JAMAAmy’s Voice of God: SGLT2i use in ADHFCCJM: https://www.ccjm.org/content/91/1/47EMPA AHF: https://pubmed.ncbi.nlm.nih.gov/38569758/Joel’s Voice of GodThe ADVOR Trial: https://www.nejm.org/doi/full/10.1056/NEJMoa2203094NephJC coverage: http://www.nephjc.com/news/advorFreely Filtered’s coverage: http://www.nephjc.com/freelyfiltered/52/advorOutline Chapter 15 — Clinical Use of DiureticsPart 2- beginning on page 460- Determinants of Diuretic responsiveness - 2 important determinants of diuretic response - Site of action - Presence of counterbalancing antinatriuretic forces - Ang2 - Aldo - Low systemic BP - Adds rate of drug excretion as # 2 and a half - Almost all diuretics are protein bound - So not well filtered - Enter tubule through organic anion and organic cation transporter - This can limit diuretic effectiveness - Natriuretic response plateaus at higher rates of diuretic excretion due to complete inhibition of the diuretic target - This plateau in normal people is 1 mg of bumetanide and 40 mg of furosemide given IV - Double this for oral furosemide, no adjustment needed for bumetanide - 15-6- Refractory edema - Start with a loop diuretic - Initial aim is to find the effective si

S1 Ep 21Chapter Fifteen: Clinical Use of Diuretics, part 1
Outline Chapter 15 — Clinical Use of Diuretics- Among most commonly used drugs- Block NaCl reabsorption at different sites along the nephron- The ability to induce negative balance has made them useful in multiple diseases- Edematous states- Hypertension- Mechanism of action- Three major classes- Loop- NaK2Cl- Up to 25% of filtered sodium excreted- Thiazide- NCC- Up to 3-5% of filtered sodium excreted- Potassium sparing- ENaC- Up to 1-2% of filtered sodium excreted- Each segment has a unique sodium channel to allow tubular sodium to flow down a concentration gradient into the cell- Table 15-1 is interesting- Most of the sodium 55-655 is reabsorbed in the proximal tubule- Proximal diuretics would be highly effective if it wasn’t for the loop and other distal sites of Na absorption- Loop Diuretics- Furosemide- Bumetanide- Torsemide- Ethacrynic acid- NaK2Cl activated when all four sites are occupied- Loop diuretic fits into the chloride slot- In addition to blocking Na reabsorption results in parallel decrease in calcium resorption- Increase in stones and nephro albinos is especially premature infants which can increase calcium excretion 10-fold- Thiazide- Even though they are less potent than loops they are great for hypertension- “Not a problem in uncomplicated hypertension where marked fluid loss is neither necessary nor desirable”- Some chlorothiazide and metolazone also inhibit carbonic anhydrase in the proximal tubule- Increase Calcium absorption. Mentions that potassium sparing diuretics do this also- Potassium sparing diuretics- Amiloride- Spironolactone- Triamterene- Act at principal cells in the cortical collecting tubule,- Block aldosterone sensitive Na channels.- Discusses the difference between amiloride and triamterene and spiro- Mentions that trimethoprim can have a similar effect- Spiro is surprisingly effective in cirrhosis and ascites- Talks about amiloride helping in lithium toxicity- Partially reverse and prevent NDI from lithium- Trial Terence as nephrotoxin?- Causes crystaluria and casts- These crystals are pH independent- Faintly radio opaque- Acetazolamide- Blocks carbonic anhydrase- Causes both NaCl and NaHCO3 loss- Modest diuresis de to distal sodium reclamation- Mannitol- Nonreabsorbable polysaccharide- Acts mostly in proximal tubule and Loop of Henle- Causes water diuresis- Was used to prevent ATN- Can cause hyperosmolality directly and through the increased water loss- This hyperosmolality will be associated with osmotic movement of water from cells resulting in hyponatremia, like in hyperglycemia.- Docs must treat the hyperosmolality not the hyponatremia- Time course of Diuresis- Efficacy of a diuretic related to- Site of action- Dietary sodium action- 15-1 shows patient with good short diuretic response but other times of low urine Na resulting in no 24 hour net sodium excretion.- Low sodium diets work with diuretics to minimize degree of sodium retension while diuretic not working- Also minimizes potassium losses- Increase frequency- Increase dose- What causes compensatory anti-diuresis- Activation of RAAS and SNS- ANG II, aldo, norepi all promote Na reabsorption- But even when prazosin to block alpha sympathetic and capto[pril to block RAAS sodium retention occurs- Decrease in BP retains sodium with reverse pressure natriuresis- Even with effective diuresis there is reestablishment of a new steady state- Diuresis is countered by- Increases in tubular reabsorption at non-diuretic sensitive sites (neurohormonal mediated)- Flow mediated in creases in Tubular reabsorption distal to the diuretic from increased sodium delivery.- Hypertrophy- Increased Na-K-ATPase activity- Decreased tubular secretion of diuretic if renal perfusion is impaired- Getting to steady state requires- Diuretic dose and sodium intake be constant- Sodium balance is reestablished with 3 days of a fixed diuretic dose- K balance in 6-9 days- Figure 15-2- Which means that people on stable doses of diuretics don’t need regular labs, the abnormalities will emerge quickly.- Maximum diuresis happens with first dose- Figure 15-3- Fluid and Electrolyte complications- Volume depletion- “Effective circulating volume depletion also can develop in patients who remain edematous. Although fluid persists, there may be a sufficient reduction in intracranial filling pressures and cardiac output to produce a clinically important reduction in tissue perfusion.”- Azotemia- Decreased effective circulating volume with diuretic therapy also can diminish renal perfusion and secondarily the GFR.- Describes the traditional reason for increased BUN:Cr ratio- Then states that as much as a third of of the rise in BUN may reflect increased urea production; it is possible, for example, that reduced skeletal muscle perfusion leads to enhanced local proteolysis. This increases urea production.- Hypokalemia- Loop and thiazide increase urinary potassium losses- Often lead to hypokalemia- 50 mg of HCTZ drop K by 0.4 to 0.6 mEq/L with 15% falling b
S1 Ep 20Chapter Fourteen: Hypovolemic States, part 2
Outline Chapter 14 — Treatment- Treatment - Both oral and IV treatment can be used for volume replacement - The goal of therapy are to restore normovolemia - And to correct associated acid-base and electrolyte disorders - Oral Therapy - Usually can be accomplished with increased water and dietary sodium - May use salt tablets - Glucose often added to resuscitation fluids - Provides calories - Promotes intestinal Na reabsorption since there is coupled Na and Glucose similar to that seen in the proximal tubule - Rice based solutions provide more calories and amino acids which also promote sodium reabsorption - 80g/L of glucose with rice vs 20 g/L with glucose alone - IV therapy - Dextrose solutions - Physiologically equivalent to water - For correcting hypernatremia - For covering insensible losses - Watch for hyperglycemia - Footnote warns against giving sterile water - Saline solutions - Most hypovolemic patients have a water and a sodium deficit - Isotonic saline has a Na concentration of 154, similar to that of plasma see page 000 - Half-isotonic saline is equivalent to 550 ml of isotonic saline and 500 of free water. Is that a typo? - 3% is a liter of hypertonic saline and 359 extra mEq of Na - Dextrose in saline solutions - Give a small amount of calories, otherwise useless - Alkalinizing solutions - 7.5% NaHCO3 in 50 ml ampules 44 mEq of Na and 44 mEq of HCO3 - Treat metabolic acidosis or hyperkalemia - Why 44 mEq and not 50? - Do not give with calcium will form insoluble CaCO3 - Polyionic solutions - Ringers contains physiologic K and Ca - Lactated Ringers adds 28 mEq of lactate - Spreads myth of LR in lactic acidosis - Potassium chloride - Available as 2 mEq/mL - Do not give as a bolus as it can cause fatal hyperkalemia - Plasma volume expanders - Albumin, polygelastins, hetastarch are restricted to vascular space - 25% albumin can pull fluid into the vascular space - 25% albumin is an albumin concentration of 25 g/dL compare to physiologic 4 g/dL - Says it pulls in several times its own volume - 5% albumin is like giving plasma - Blood - Which fluid? - Look at osmolality, give hypotonic fluids to people with high osmolality - Must include all electrolytes - Example of adding 77 mEw of K to 0.45 NS and making it isotonic - DI can be replaced with dextrose solutions, pure water deficit - Case 14-3 - Diarrhea with metabolic acidosis - He chooses 0.25 NS with 44 mEq of NaCl and 44 NaHCO3 - Talks about blood and trauma - Some studies advocate delaying saline until penetrating trauma is corrected APR about to. Keep BP low to prevent bleeding. Worry about diluting coagulation factors - Only do this if the OR is quickly available - Volume deficit - Provides formula for water deficit and sodium deficit - Do not work for isotonic losses - Provides a table to adjust fluid loss based on changes in Hgb or HCTZ - Says difficult to estimate it from lab findings and calculations - Follow serial exams - Serial urine Na - Rate of replacement - Goal is not to give fluid but to induce a positive balance - Suggests 50-100 ml/hr over what is coming out of the body - Urine - Insensibles 30-50 - Diarrhea - Tubes - Hypovolemic shock - Due to bleeding - Sequesting in third space - Why shock? - Progressive volume depletion leads to - Increased sympathetic NS - Increased Ang 2 - Initially this maintains BP, cerebral and coronary circulation - But this can decrease splanchnic, renal and mucocutaneous perfusion - This leads to lactic acicosis - This can result in intracellular contents moving into circulation or translocation of gut bacteria - Early therapy to prevent irreversible shock - In dogs need to treat with in 2 hours - In humans may need more than 4 hours - Irreversible shock associated with pooling of blood in capillaries - Vasomotor paralysis - Hyperpolarization of vascular smooth muscle as depletion of ATP allows K to flowing out from K channels opening. Ca flows out too leading to vasodilation - Glyburide is an K-ATP channel inhibitor (?) caused increased vasoconstriction and BP - Pluggin of capillaries by neutrophils - Cerebral ischemia - Increased NO generation - Which Fluids? - Think of what is lost and replace that. - Bleeding think blood - Raise the hct but not above 35 - Acellular blood substitutes, looked bad at the time of this writing - Di aspirin cross linked hemoglobin had increased 2 and 28 day mortality vs saline - Colloids sound great but they fail in RCTs - SAFE - FEAST - Points out that saline replaces the interstitial losses why do we think those losses are unimportant - Pulmonary circulation issue - Pulmonary circulation is more leaky so oncotic pressure less effective there - Talks about the lungs be naturally protected from pulmonary edema - Rate of fluid - 1-2 liters in first hour - Suggests CVP or capillary wedge pressure during resuscitation - No refs in the rate of fluid administration section - Lactic acidosis - Points out that HCO can impair lactate utilization - Also states that arterial p

S1 Ep 19Chapter Fourteen: Hypovolemic States, part 1
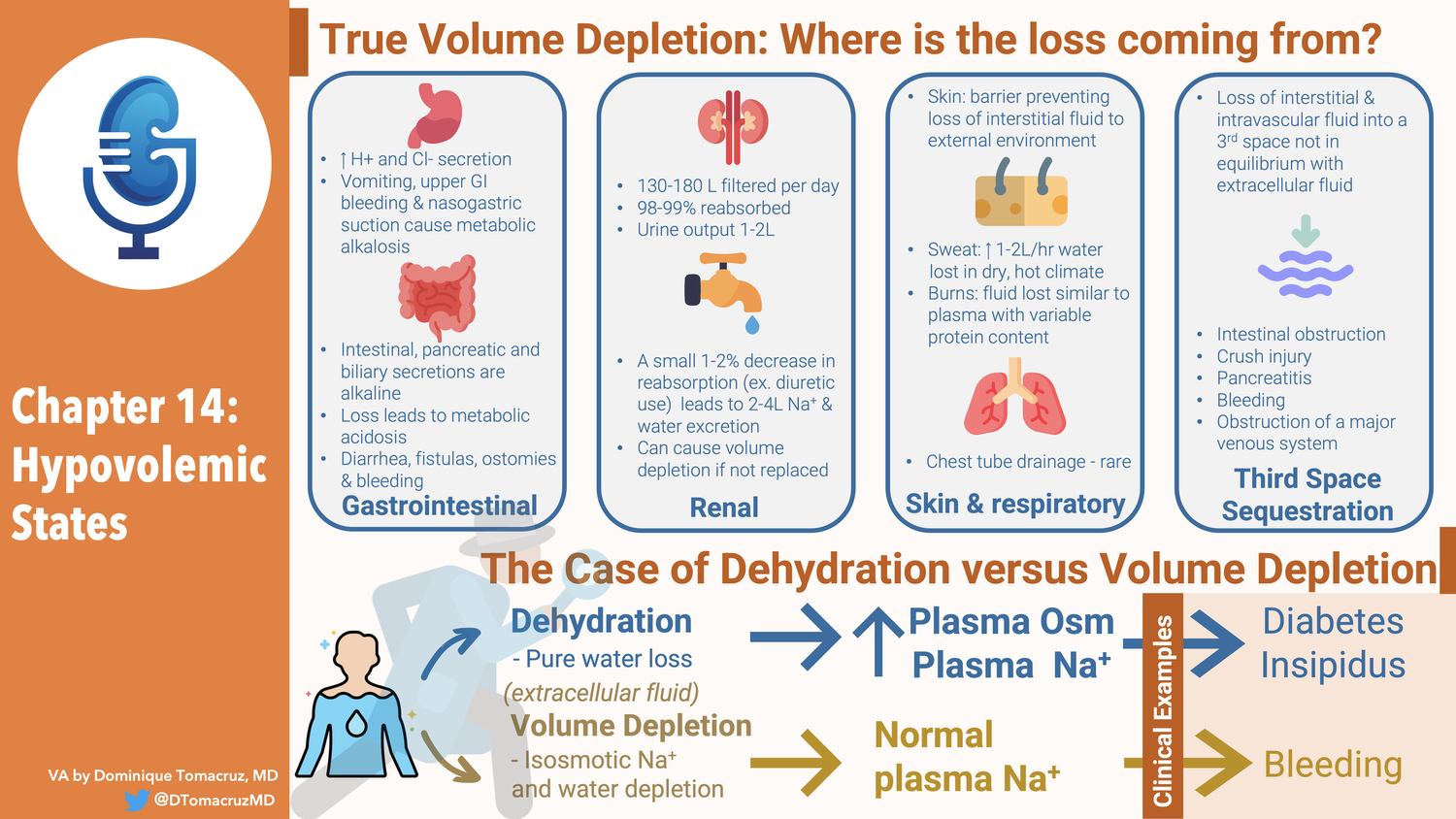
OutlineChapter 14- Hypovolemic States- Etiology - True volume depletion occurs when fluid is lost from from the extracellular fluid at a rate exceeding intake - Can come the GI tract - Lungs - Urine - Sequestration in the body in a “third space” that is not in equilibrium with the extracellular fluid. - When losses occur two responses ameliorate them - Our intake of Na and fluid is way above basal needs - This is not the case with anorexia or vomiting - The kidney responds by minimizing further urinary losses - This adaptive response is why diuretics do not cause progressive volume depletion - Initial volume loss stimulates RAAS, and possibly other compensatory mechanisms, resulting increased proximal and collecting tubule Na reabsorption. - This balances the diuretic effect resulting in a new steady state in 1-2weeks - New steady state means Na in = Na out - GI Losses - Stomach, pancreas, GB, and intestines secretes 3-6 liters a day. - Almost all is reabsorbed with only loss of 100-200 ml in stool a day - Volume depletion can result from surgical drainage or failure of reabsorption - Acid base disturbances with GI losses - Stomach losses cause metabolic alkalosis - Intestinal, pancreatic and biliary secretions are alkalotic so losing them causes metabolic acidosis - Fistulas, laxative abuse, diarrhea, ostomies, tube drainage - High content of potassium so associated with hypokalemia - [This is a mistake for stomach losses] - Bleeding from the GI tract can also cause volume depletion - No electrolyte disorders from this unless lactic acidosis - Renal losses - 130-180 liters filtered every day - 98-99% reabsorbed - Urine output of 1-2 liters - A small 1-2% decrease in reabsorption can lead to 2-4 liter increase in Na and Water excretion - 4 liters of urine output is the goal of therapeutic diuresis which means a reduction of fluid reabsorption of only 2% - Diuretics - Osmotic diuretics - Severe hyperglycemia can contribute to a fluid deficit of 8-10 Iiters - CKD with GFR < 25 are poor Na conservers - Obligate sodium losses of 10 to 40 mEq/day - Normal people can reduce obligate Na losses down to 5 mEq/day - Usually not a problem because most people eat way more than 10-40 mEq of Na a day. - Salt wasting nephropathies - Water losses of 2 liters a day - 100 mEq of Na a day - Tubular and interstitial diseases - Medullary cystic kidney - Mechanism - Increased urea can be an osmotic diuretic - Damage to tubular epithelium can make it aldo resistant - Inability to shut off natriuretic hormone (ANP?) - The decreased nephro number means they need to be able to decrease sodium reabsorption per nephron. This may not be able to be shut down acutely. - Experiment, salt wasters can stay in balance if sodium intake is slowly decreased. (Think weeks) - Talks about post obstruction diuresis - Says it is usually appropriate rather than inappropriate physiology. - Usually catch up solute and water clearance after releasing obstruction - Recommends 50-75/hr of half normal saline - Talks briefly about DI - Skin and respiratory losses - 700-1000 ml of water lost daily by evaporation, insensible losses (not sweat) - Can rise to 1-2 liters per hour in dry hot climate - 30-50 mEq/L Na - Thirst is primary compensation for this - Sweat sodium losses can result in hypovolemia - Burns and exudative skin losses changes the nature of fluid losses resulting in fluid losses more similar to plasma with a variable amount of protein - Bronchorrhea - Sequestration into a third space - Volume Deficiency produced by the loss of interstitial and intravascular fluid into a third space that is not in equilibrium with the extracellular fluid. - Hip fracture 1500-2000 into tissues adjacent to fxr - Intestinal obstruction, severe pancreatitis, crush injury, bleeding, peritonitis, obstruction of a major venous system - Difference between 3rd space and cirrhosis ascities - Rate of accumulation, if the rate is slow enough there is time for renal sodium and water compensation to maintain balance. - So cirrhotics get edema from salt retension and do not act as hypovolemia - Hemodynamic response to volume depletion - Initial volume deficit reduced venous return to heart - Detected by cardiopulmonary receptors in atria and pulmonary veins leading to sympathetic vasoconstriction in skin and skeletal muscle. - More marked depletion will result in decreased cardiac output and decrease in BP - This drop in BP is now detected by carotid and aortic arch baroreceptors resulting in splanchnic and renal circulation vasoconstriction - This maintains cardiac and cerebral circulation - Returns BP toward normal - Increase in BP due to increased venous return - Increased cardiac contractility and heart rate - Increased vascular resistance - Sympathetic tone - Renin leading to Ang2 - These can compensate for 500 ml of blood loss (10%) - Unless there is autonomic dysfunction - With 16-25% loss this will not compensate for BP when patient upright - Postural dizziness - Sy

S1 Ep 18Chapter Thirteen: Meaning and Application of Urine Chemistries
ReferencesJC mentioned that the diagnostic accuracy of 24 hour urine collection increases with more collections! Metabolic evaluation of patients with recurrent idiopathic calcium nephrolithiasisWe didn't refer to a particular study on sodium intake and the 24 hour urine but this meta-analysis Comparison of 24‐hour urine and 24‐hour diet recall for estimating dietary sodium intake in populations: A systematic review and meta‐analysis - PMC 24‐hour diet recall underestimated population mean sodium intake.Anna looking up ace i and urinary sodium Effects of ACE inhibition on proximal tubule sodium transport | American Journal of Physiology-Renal PhysiologyThe original FENa paper by Espinel: The FeNa Test: Use in the Differential Diagnosis of Acute Renal Failure | JAMA | JAMA NetworkSchreir’s replication and expansion of Espinel’s data: Urinary diagnostic indices in acute renal failure: a prospective studyHere’s a report from our own JC on the Diagnostic Utility of Serial Microscopic Examination of the Urinary Sediment in Acute Kidney Injury | American Society of NephrologyJC shared his journey regarding FENa and refers to his recent paper Concomitant Identification of Muddy Brown Granular Casts and Low Fractional Excretion of Urinary Sodium in AKIAnd Melanie’s accompanying editorial Mind the Cast: FENa versus Microscopy in AKI : Kidney360 (with a great image from Samir Parikh)JC referenced this study from Schrier on FENa with a larger series: Urinary diagnostic indices in acute renal failure: a prospective studyNonoliguric Acute Renal Failure Associated with a Low Fractional Excretion of Sodium | Annals of Internal MedicineUrine sodium concentration to predict fluid responsiveness in oliguric ICU patients: a prospective multicenter observational study | Critical Care | Full TextA classic favorite: Acute renal success. The unexpected logic of oliguria in acute renal failure Marathon runners had granular casts in their urine without renal failure. Kidney Injury and Repair Biomarkers in Marathon RunnersCute piece from Rick Sterns on urine electrolytes! Managing electrolyte disorders: order a basic urine metabolic panelThe Urine Anion Gap: Common Misconceptions | American Society of NephrologyThe urine anion gap in context CJASNExcellent review from Halperin on urine chemistries (including some consideration of the TTKG): Use of Urine Electrolytes and Urine Osmolality in the Clinical Diagnosis of Fluid, Electrolytes, and Acid-Base Disorders - Kidney International ReportsRenal tubular acidosis (RTA): Recognize The Ammonium defect and pHorget the urine pH | SpringerLinkOutlineChapter 13- New part: Part 3, Physiologic approach to acid-base and electrolyte disorders - Do you remember the previous two parts? - Renal physiology - Regulation of water and electrolyte balance- Chapter 13: Meaning and application of urine chemistries - Measurement of urinary electrolyte concentrations, osmolality and pH helps diagnose some conditions - There are no fixed normal values - Kidney varies rate of excretion to match intake and endogenous production - Example: urine Na of 125/day can be normal if patient euvolemic on a normal diet, and wildly inappropriate in a patient who is volume depleted. - Urine chemistries are: - Useful - Simple - Widely available - Usually a random sample is adequate - 24-hour samples give additional context - Gives example of urinary potassium, with extra renal loss of K, urine K should be < 25, but if the patient has concurrent volume deficiency and urine output is only 500 mL, then urine K concentration can appropriately be as high as 40 mEq/L - Table 13-1 - Seems incomplete, see my notes on page 406 - What is Gravity ARF?- Sodium Excretion - Kidney varies Na to maintain effective circulating volume (I’d say volume homeostasis) - Urine Na affected by RAAS and ANP - Na concentration can be used to determine volume status - Urine Na < 20 is hypovolemia - Says it is especially helpful in determining the etiology of hyponatremia - Calls out SIADH and volume depletion - Used 40 mEq/L for SIADH - Also useful in AKI - Where differential is pre-renal vs ATN - In addition to urine Na (and FENa) look at urine osmolality - Again uses 40 mEq/l - Mentions FENa and urine osmolality - Urine Na can estimate dietary sodium intake - Suggests doing this during treatment of hypertension to assure dietary compliance - 24 hour urine Na is accurate with diuretics as long as the dose is stable and the drugs are chronic - Diuretics increase Na resorption in other segments of the tubule that are not affected by the diuretic - Points to increased AT2 induced proximal Na resorption and aldosterone induced DCT resoprtion - In HTN shoot for less than 100 mEq/Day - Urine Na useful in stones - Urine uric acid and urine Ca can cause stones and their handling is dependent on sodium - Low sodium diet can mask elevated excretion of these stone forming metabolites - 24-hour Na > 75 and should be enough sodium to avoid this p

S1 Ep 16Chapter Eleven: Regulation of Acid-Base Balance, part 2
ReferencesWe considered the complexity of the machinery to excrete ammonium in the context of research on dietary protein and how high protein intake may increase glomerular pressure and contribute to progressive renal disease (many refer to this as the “Brenner hypothesis”). Dietary protein intake and the progressive nature of kidney disease: the role of hemodynamically mediated glomerular injury in the pathogenesis of progressive glomerular sclerosis in aging, renal ablation, and intrinsic renal diseaseA trial that studied low protein and progression of CKD The Effects of Dietary Protein Restriction and Blood-Pressure Control on the Progression of Chronic Renal Disease(and famously provided data for the MDRD eGFR equation A more accurate method to estimate glomerular filtration rate from serum creatinine: a new prediction equation. Modification of Diet in Renal Disease Study GroupWe wondered about dietary recommendations in CKD. of note, this is best done in the DKD guidelines from KDIGO Executive summary of the 2020 KDIGO Diabetes Management in CKD Guideline: evidence-based advances in monitoring and treatment.Joel mentioned this study on red meat and risk of ESKD. Red Meat Intake and Risk of ESRDWe referenced the notion of a plant-based diet. This is an excellent review by Deborah Clegg and Kathleen Hill Gallant. Plant-Based Diets in CKD : Clinical Journal of the American Society of NephrologyHere’s the review that Josh mentioned on how the kidney appears to sense pH Molecular mechanisms of acid-base sensing by the kidneyRemarkably, Dr. Dale Dubin put a prize in his ECG book Free Car Prize Hidden in Textbook Read the fine print: Student wins T-birdA review of the role of the kidney in DKA: Diabetic ketoacidosis: Role of the kidney in the acid-base homeostasis re-evaluatedJosh mentioned the effects of infusing large amounts of bicarbonate The effect of prolonged administration of large doses of sodium bicarbonate in man and this study on the respiratory response to a bicarbonate infusion: The Acute Effects In Man Of A Rapid Intravenous Infusion Of Hypertonic Sodium Bicarbonate Solution. Ii. Changes In Respiration And Output Of Carbon DioxideThis is the study of acute respiratory alkalosis in dogs: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC293311/?page=1And this is the study of medical students who went to the High Alpine Research Station on the Jungfraujoch in the Swiss Alps https://www.nejm.org/doi/full/10.1056/nejm199105163242003Self explanatory! A group favorite! It Is Chloride Depletion Alkalosis, Not Contraction AlkalosisEffects of chloride and extracellular fluid volume on bicarbonate reabsorption along the nephron in metabolic alkalosis in the rat. Reassessment of the classical hypothesis of the pathogenesis of metabolic alkalosisA review of pendrin’s role in volume homeostasis: The role of pendrin in blood pressure regulation | American Journal of Physiology-Renal PhysiologyInfusion of bicarbonate may lead to a decrease in respiratory stimulation but the shift of bicarbonate to the CSF may lag. Check out this review Neural Control of Breathing and CO2 Homeostasis and this classic paper Spinal-Fluid pH and Neurologic Symptoms in Systemic Acidosis.OutlineOutline: Chapter 11- Regulation of Acid-Base Balance- Introduction - Bicarb plus a proton in equilibrium with CO2 and water - Can be rearranged to HH - Importance of regulating pCO2 and HCO3 outside of this equation - Metabolism of carbs and fats results in the production of 15,000 mmol of CO2 per day - Metabolism of protein and other “substances” generates non-carbonic acids and bases - Mostly from sulfur containing methionine and cysteine - And cationic arginine and lysine - Hydrolysis of dietary phosphate that exists and H2PO4– - Source of base/alkali - Metabolism of an ionic amino acids - Glutamate and asparatate - Organic anions going through gluconeogenesis - Glutamate, Citrate and lactate - Net effect on a normal western diet 50-100 mEq of H+ per day - Homeostatic response to these acid-base loads has three stages: - Chemical buffering - Changes in ventilation - Changes in H+ excretion - Example of H2SO4 from oxidation of sulfur containing AA - Drop in bicarb will stimulate renal acid secretion - Nice table of normal cid-base values, arterial and venous- Great 6 bullet points of acid-base on page 328 - Kidneys must excrete 50-100 of non-carbonic acid daily - This occurs by H secretion, but mechanisms change by area of nephron - Not excreted as free H+ due to minimal urine pH being equivalent to 0.05 mmol/L - No H+ can be excreted until virtually all of th filtered bicarb is reabsorbed - Secreted H+ must bind buffers (phosphate, NH3, cr) - PH is main stimulus for H secretion, though K, aldo and volume can affect this.- Renal Hydrogen excretion - Critical to understand that loss of bicarb is like addition of hydrogen to the body - So all bicarb must be reabsorbed before dietary H load can be secreted - GFR of 125 and bicarb of 24 resu

S1 Ep 15The 2023 NKF Clinical Meeting Live Recording: Diuretic Draft
The Channelers went where no nephrology podcasters have gone before, recording in front of a live audience at the National Kidney Foundation Clinical Meeting in Austin. We had all eight Channelers doing a live podcast.We did a Freely Filtered-inspired draft of the best diuretics.The draft order:Leticia Rolon Anna Gaddy Joel TopfRoger Rodby Josh Waitzman Amy Yau JC Velez And Melanie HoenigReferencesJC Tolvaptan in Later-Stage Autosomal Dominant Polycystic Kidney DiseaseIntravenous conivaptan for the treatment of hyponatraemia caused by the syndrome of inappropriate secretion of antidiuretic hormone in hospitalized patients: a single-centre experienceRapidity of Correction of Hyponatremia Due to Syndrome of Inappropriate Secretion of Antidiuretic Hormone Following TolvaptanTolvaptan, a Selective Oral Vasopressin V2-Receptor Antagonist, for HyponatremiaJosh Review on amiloride development https://pubmed.ncbi.nlm.nih.gov/7039345/Toad bladder: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC1351665/Amiloride derivatives that inhibit flagella: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC8544414/Amiloride as taste sensor: https://www.science.org/doi/10.1126/science.6691151Batlle on diabetes Insipidus: https://www.nejm.org/doi/full/10.1056/NEJM198502143120705?query=recirc_curatedRelated_articleAmiloride + ddavp for DI https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2518801/Amy Treatment of refractory congestive heart failure and normokalemic hypochloremic alkalosis with acetazolamide and spironolactone.Acetazolamide reversibly inhibits water conduction by aquaporin-4Inhibition of Human Aquaporin-1 Water Channel Activity by Carbonic Anhydrase InhibitorsAcetazolamide Attenuates Lithium-Induced Nephrogenic Diabetes InsipidusAcetazolamide in Lithium-Induced Nephrogenic Diabetes InsipidusIn Vivo Antibacterial Activity of AcetazolamideRoger50th anniversary of aldosteroneJoelSotagliflozin in Patients with Diabetes and Recent Worsening Heart FailureThe SGLT2 inhibitor empagliflozin in patients hospitalized for acute heart failure: a multinational randomized trialEffects of Early Empagliflozin Initiation on Diuresis and Kidney Function in Patients With Acute Decompensated Heart Failure (EMPAG-HF)Empagliflozin and Heart failure: Diuretic and Cardiorenal EffectsAnnaClinical Results of Treatment of Diabetes Insipidus with Drugs of the Chlorothiazide SeriesTreatment of nephrogenic diabetes insipidus with hydrochlorothiazide and amilorideInfluence of renal nerves and sodium balance on the acute antidiuretic effect of bendroflumethiazide in rats with diabetes insipidusAntidiuretic effect of hydrochlorothiazide in lithium-induced nephrogenic diabetes insipidus is associated with upregulation of aquaporin-2, Na-Cl co-transporter, and epithelial sodium channelMajor Outcomes in High-Risk Hypertensive Patients Randomized to Angiotensin-Converting Enzyme Inhibitor or Calcium Channel Blocker vs DiureticWalsh AC, Moyes A. Intractable Congestive Heart Failure Successfully Treated With Southey Tubes. Can Med Assoc J. 1964 Jun 13;90(24):1375-6.Godwin TF, Gunton RW. Clinical trial of a new diuretic, furosemide: comparison with hydrochlorothiazide and mercaptomerin. Can Med Assoc J. 1965 Dec 18;93(25):1296-300.Gerber JG. Role of prostaglandins in the hemodynamic and tubular effects of furosemide. Fed Proc. 1983 Apr;42(6):1707-10.Schlatter E, Salomonsson M, Persson AE, Greger R. Macula densa cells sense luminal NaCl concentration via furosemide sensitive Na+2Cl-K+ cotransport. Pflugers Arch. 1989 Jul;414(3):286-90. doi: 10.1007/BF00584628.
S1 Ep 14A Very Special Episode: Meet the Glaucomfleckens

S1 Ep 13Chapter Eleven: Regulation of Acid-Base Balance, part 1
ReferencesWe considered the effect of a high protein diet and potential metabolic acidosis on kidney function. This review is of interest by Donald Wesson, a champion for addressing this issue and limiting animal protein: Mechanisms of Metabolic Acidosis-Induced Kidney Injury in Chronic Kidney DiseaseHostetter explored the effect of a high protein diet in the remnant kidney model with 1 ¾ nephrectomy. Rats with reduced dietary acid load (by bicarbonate supplementation) had less tubular damage. Chronic effects of dietary protein in the rat with intact and reduced renal massWesson explored treatment of metabolic acidosis in humans with stage 3 CKD in this study. Treatment of metabolic acidosis in patients with stage 3 chronic kidney disease with fruits and vegetables or oral bicarbonate reduces urine angiotensinogen and preserves glomerular filtration rateIn addition to the effect of metabolic acidosis from a diet high in animal protein, this diet also leads to hyperfiltration. This was demonstrated in normal subjects; ingesting a protein diet had a significantly higher creatinine clearance than a comparable group of normal subjects ingesting a vegetarian diet. Renal functional reserve in humans: Effect of protein intake on glomerular filtration rate.This finding has been implicated in Brenner’s theory regarding hyperfiltration: The hyperfiltration theory: a paradigm shift in nephrologyOne of multiple publications from Dr. Nimrat Goraya whom Joel mentioned in the voice over: Dietary Protein as Kidney Protection: Quality or Quantity?We wondered about the time course in buffering a high protein meal (and its subsequent acid load on ventilation) and Amy found this report:Effect of Protein Intake on Ventilatory Drive | Anesthesiology | American Society of Anesthesiologists Roger mentioned that the need for acetate to balance the acid from amino acids in parenteral nutrition was identified in pediatrics perhaps because infants may have reduced ability to generate acid. Randomised controlled trial of acetate in preterm neonates receiving parenteral nutrition - PMCHe also recommended an excellent review on the complications of parenteral nutrition by Knochel https://www.kidney-international.org/action/showPdf?pii=S0085-2538%2815%2933384-6 which explained that when the infused amino acids disproportionately include cationic amino acids, metabolism led to H+ production. This is typically mitigated by preparing a solution that is balanced by acetate. Amy mentioned this study that explored the effect of protein intake on ventilation: Effect of Protein Intake on Ventilatory Drive | Anesthesiology | American Society of AnesthesiologistsAnna and Amy reminisced about a Skeleton Key Group Case from the renal fellow network Skeleton Key Group: Electrolyte Case #7JC wondered about isolated defects in the proximal tubule and an example is found here: Mutations in SLC4A4 cause permanent isolated proximal renal tubular acidosis with ocular abnormalitiesAnna’s Voiceover re: Gastric neobladder → metabolic alkalosis and yes, dysuria. The physiology of gastrocystoplasty: once a stomach, always a stomach but not as common as you might think Gastrocystoplasty: long-term complications in 22 patientsSjögren’s syndrome has been associated with acquired distal RTA and in some cases, an absence of the H+ ATPase, presumably from autoantibodies to this transporter. Here’s a case report: Absence of H(+)-ATPase in cortical collecting tubules of a patient with Sjogren's syndrome and distal renal tubular acidosisCan't get enough disequilibrium pH? Check this out- Spontaneous luminal disequilibrium pH in S3 proximal tubules. Role in ammonia and bicarbonate transport.Acetazolamide secretion was studied in this report Concentration-dependent tubular secretion of acetazolamide and its inhibition by salicylic acid in the isolated perfused rat kidney. | Drug Metabolism & DispositionIn this excellent review, David Goldfarb tackles the challenging case of a A Woman with Recurrent Calcium Phosphate Kidney Stones (spoiler alert, many of these patients have incomplete distal RTA and this problem is hard to treat). Molecular mechanisms of renal ammonia transport excellent review from David Winer and Lee Hamm. OutlineOutline: Chapter 11- Regulation of Acid-Base Balance- Introduction - Bicarb plus a proton in equilibrium with CO2 and water - Can be rearranged to HH - Importance of regulating pCO2 and HCO3 outside of this equation - Metabolism of carbs and fats results in the production of 15,000 mmol of CO2 per day - Metabolism of protein and other “substances” generates non-carbonic acids and bases - Mostly from sulfur containing methionine and cysteine - And cationic arginine and lysine - Hydrolysis of dietary phosphate that exists and H2PO4– - Source of base/alkali - Metabolism of an ionic amino acids - Glutamate and asparatate - Organic anions going through gluconeogenesis - Glutamate, Citra
S1 Ep 12Chapter Ten: Acid-Base Physiology
References for Chapter 10We did not mention many references in our discussion today but our listeners may enjoy some of the references below. Effects of pH on Potassium: New Explanations for Old Observations - PMC although the focus of this article is on potassium, this elegant review by Aronson and Giebisch reviews intracellular shifts as it relates to pH and K+.Josh swooned for Figure 10-1 is this right? Which figure was it? which shows the relationship between [H+] and pH. You can find this figure in the original reference from Halperin ML and others, Figure 1 here. Factors That Control the Effect of pH on Glycolysis in Leukocytes Here’s Leticia Rolon’s favorite Henderson-Hasselbalch calculator website: Henderson-Hasselbalch Calculator | Buffer Solutions [hint! for this site, use the bicarbonate (or “total CO2”) for A- and PCO2 for the HA] There’s also a cooking tab for converting units! Fundamentals of Arterial Blood Gas Interpretation - PMC this review published posthumously from the late but beloved Jerry Yee and his group at Henry Ford Hospital, explores the details and underpinnings of our understandings of arterial blood gas interpretation (and this also addresses how our colleagues in clinical chemistry measure total CO2 - which JC referenced- but JC said “machine” and our colleagues prefer the word “instrument.”)Amy went deep on bicarbonate in respiratory acidosis. Here are her refs:Sodium bicarbonate therapy for acute respiratory acidosisSodium Bicarbonate in Respiratory AcidosisBicarbonate therapy in severe metabolic acidosisEffect of Intravenous Sodium Bicarbonate on Ventilation, Gas Exchange, and Acid-Base Balance in Patients with Chronic Pulmonary InsufficiencyBicarbonate Therapy in Severe Metabolic Acidosis | American Society of Nephrology this review article from Sabatini and Kurtzman addresses the issues regarding bicarbonate therapy including theoretical intracellular acidosis. Bicarbonate in DKA? Don’t do it: Bicarbonate in diabetic ketoacidosis - a systematic review Here’s a review from Bushinsky and Krieger on the effect acidosis on bone https://www.sciencedirect.com/science/article/abs/pii/S0085253822002174Here is the primary resource that Anna used in here investigation of meat replacements Nutritional Composition of Novel Plant-Based Meat Alternatives and Traditional Animal-Based MeatsWe enjoyed this paper that Dr. Rose references from the Journal of Clinical Investigation 1955 in which investigators infused HCl into nephrectomized dogs and observed changes in extracellular ions. https://www.jci.org/articles/view/103073/pdWe wondered about the amino acids/protein in some available meat alternatives they are explored in this article in the journal Amino Acids: Protein content and amino acid composition of commercially available plant-based protein isolates - PMC and you may enjoy this exploration of the nutritional value of these foods: Full article: Examination of the nutritional composition of alternative beef burgers available in the United StatesOutlineChapter 10: Acid-Base Physiology - H concentration regulated tightly - Normal H+ is 40 nm/L - This one millionth the concentration of Na and K - It needs to be this dilute because H+ fucks shit up - Especially proteins - Cool foot note H+ actually exists as H3O+ - Under normal conditions the H+ concentration varies little from normal due to three steps - Chemical buffering by extracellular and intracellular bufffers - Control of partial pressure of CO2 by alterations of alveolar ventilation - Control of plasma bicarbonate by changes in renal H+ excretion - Acid and bases - Use definition by Bronsted - Acid can donate protons - Base can accept protons - There are two classes of acids** - Carbonic acid H2CO3 - Each day 15000 mmol of CO2 are generated - CO2 not acid but combines with water to form carbonic acid H2CO3 - CO2 cleared by the lungs - Noncarbonic acid - Formed from metabolism of protein. Sulfur containing AA generate H2SO4. Only 50-100 mEq of acid produced from these sources. - Cleared by the kidneys - Law of Mass Action - Velocity of reaction proportional to the product of the concentrations of the reactants - Goes through mass action formula for water - Concludes that water has H of 155 nanoM/L, more than the 40 in plasma - Says you can do the same mass experiment for every acid in the body - Can do it also for bases but he is not going to. - Acids and Bases can be strong or weak - Strong acids completely dissociate - Weak acids not so much - H2PO4 is only 80% dissociated - Weak acids are the principle buffers in the body - Then he goes through how H is measured in the blood and it becomes clear why pH is a logical way to measure. - Then there is a lot of math - HH equation - Derives it - Then uses it to look at phos. Very interesting application - Buffers - Goes tot he phosphate well again. Amazing math describing how powerful buffers can be - Big picture the closer the pKa is to the starting
S1 Ep 11Chapter Nine: Regulation of Plasma Osmolality
References for Chapter 9One of the few papers that Rose wrote as a single author explores electrolyte free water clearance. This seminal paper explores the issue in greater detail than the book. A New approach to disturbances in the plasma sodium concentrationWondering about the volume of sweat? Josh taught us that the volume of “transepidermal volume loss” is not affected by humidity https://www.jidonline.org/article/S0022-202X(15)48145-X/pdf but is greatly affected by temperature: Skin temperature and transepidermal water lossRegarding normal sweat physiology, there is a nice review (with figures!) titled Physiological mechanisms determining sweat composition which describes all the important cells and channels which make up sweat glands. And an important follow on paper titled Higher Bioelectric Potentials due to Decreased Chloride Absorption in the Sweat Glands of Patients with Cystic Fibrosis describing specifically the sweat characteristics of patients with cystic fibrosis.Melanie was enchanted by work from RA McCance who did early experiments to induce sodium deficiency using very low sodium diets and a homemade sauna-like tent. His musings are fascinating. Lancet 1936 Experimental human salt deficiency MEDICAL PROBLEMS IN MINERAL METABOLISMAge-related decline in urine concentration may not be universal: Comparative study from the US and two small-scale societies from Jeff Sands (of urea transport fame!)In this initial report, after continually water loading 21 volunteers, the younger group (mean age 31) had a urine osmolality of 52 mOsm/kg compared to in the older group (mean age 84). Influence of age, renal disease, hypertension, diuretics, and calcium on the antidiuretic responses to suboptimal infusions of vasopressin. In a later report older subjects (mean age 72) vs younger controls (mean age 26) drank 20 ml/kg over 40 minutes. The younger group excreted more of the water in the first 2 hours and had a lower mean urine osmolality 86 vs 112 mOsm/kg compared to the older participants. Age-associated Alterations in Thirst and Arginine Vasopressin in Response to a Water or Sodium Load Howard Furst suggests the urine to plasma electrolyte ratio as a simpler strategy to consider the free water clearance: https://nephrology.edublogs.org/files/2014/03/Water-Restriction-in-Hyponatremia1-1eb8n40.pdf or via pubmed: The urine/plasma electrolyte ratio: a predictive guide to water restrictionRapidity of Correction of Hyponatremia Due to Syndrome of Inappropriate Secretion of Antidiuretic Hormone Following TolvaptanInfoSnack picture of pre and post tolvaptan